Clean meat
By Laura Cassiday
February 2018
- Clean meat—also known as lab-grown, in vitro, or cultured meat—is meat that is grown in cell culture, rather than in an animal’s body.
- Potential benefits of clean meat include sustainability, environmental friendliness, animal welfare, food safety, and novel foods.
- Technological challenges include scaling up the production process, reducing cost, optimizing serum-free culture media, producing structured meats, and adding fat.
- Consumer acceptance of clean meat is still uncertain.
In 1932, in a collection of essays entitled Thoughts and Adventures, British statesman Winston Churchill made a bold prediction: “Fifty years hence, we shall escape the absurdity of growing a whole chicken in order to eat the breast or wing, by growing these parts separately under a suitable medium.” Although this idea may still seem like science fiction, in recent years several companies have made great strides in developing lab-grown or “clean” meat. If the technology reaches its full potential, clean meat could help satisfy the planet’s growing demand for protein, and also provide benefits for the environment, animal welfare, and food safety.
By 2050, worldwide meat consumption will increase by 73%, according to a 2011 United Nations Food and Agriculture Organization (FAO) report (http://tinyurl.com/FAO-livestock2). This surge is driven by the expanding world population, as well as by increased meat consumption in developing countries. The livestock sector already consumes about 70% of global agricultural land, divided between animal grazing and feed crops (FAO, 2009, http://tinyurl.com/FAO-livestock1). Thus, under the current system of livestock production, not enough land will be available to satisfy the growing demand for meat, and meat will become a scarce, expensive luxury item.
An obvious solution would be to substitute plant- or insect-based proteins for meat in the human diet. However, humans have consumed meat since the dawn of the species’ evolution (Larsen, C. S., J. Nutr. 133: 3893S-3897S), and meat is a rich component of numerous culinary traditions. Therefore, many people crave meat and find vegetarian diets difficult to follow. Vegetarian foods that mimic meat (e.g., veggie burgers and meatballs) have been introduced, but these products have so far failed to exactly replicate the complex flavor and textural profiles of real meat. Although many challenges still need to be overcome, clean meat may be a solution to the world’s looming protein shortage that allows people to maintain their dietary preferences.
First bites
Clean meat is meat that is grown in cell culture, rather than inside animals. Also known as cultured meat, in vitro meat, or lab-grown meat, “clean meat” is the term preferred by proponents because the meat is reportedly cleaner than meat from slaughtered animals in terms of both sanitation and environmental friendliness (Friedrich, B., http://tinyurl.com/clean-meat, 2016). The term “clean” may also refer to the conscience of the consumer because no animals must be slaughtered to produce the meat. Instead, stem cells are removed from an animal by a harmless biopsy and then cultivated in vitro to form muscle fibers.
The in vitro cultivation of muscle fibers was reported as early as 1971, when a researcher grew guinea pig aortic smooth muscle in Petri dishes (Ross, R., http://dx.doi.org/10.1083/jcb.50.1.172). In the 1990s, the US National Aeronautics and Space Administration (NASA) took interest in clean meat as a possible protein source for astronauts during long space voyages. In 2002, a NASA-funded research project produced fish “filets” from goldfish skeletal muscle explants that were expanded in vitro (Benjaminson, M. A., et al., https://doi.org/10.1016/S0094-5765(02)00033-4). The filets, which were fried and breaded, were judged by a food panel to resemble and smell like real fish filets, although US Food and Drug Administration regulations prevented the panelists from actually tasting the experimental food.
In 2004, the Dutch government began funding research on cultured meat. The project focused on three areas: stem cell biology, tissue engineering, and culture media. Although the grant ended in 2009, Maastricht University professor Mark Post continued his research on cultured meat, funded by Google co-founder Sergey Brin. On August 6, 2013, Post and his colleagues presented the first lab-grown burger at a news conference in London. The researchers produced the burger by removing stem cells from the shoulder muscle of a cow, growing them into thin strips of muscle in tissue culture flasks, and combining about 20,000 strips to make a burger.
The lab-grown burger was seasoned and cooked in butter by a chef and tasted by Post, food writer Josh Schonwald, and food researcher Hanni Rützler (Fig. 1). “I know there is no fat in it, so I didn’t really know how juicy it would be, but there is quite some intense taste,” said Rützler. “It’s close to meat, it’s not that juicy, but the consistency is perfect. This is meat to me” (Hogenboom, M., http://tinyurl.com/bbc-meat, 2016). The cost to make the burger: €250,000 (about $330,000 USD). Post is now chief scientific officer of a Maastricht University spin-off company called Mosa Meat (Maastricht, The Netherlands), which plans to scale up and commercialize the technology. Post estimates that their cultured beef will likely be introduced to high-end restaurants and specialty stores in 3–4 years, and to supermarkets after another 2–3 years.
FIG. 1. The world's first lab-grown hamburger, presented at a news conference in London in August 2013
Credit: David Parry/PA Wire
In March 2016, Memphis Meats, located in San Francisco, California, USA, unveiled the first lab-grown meatball, at a cost of $18,000 USD per pound. A year later, the company rolled out samples of clean chicken nuggets and duck à l’orange (Fig. 2). By this time, the cost had dropped to $6,000 per pound. The company’s target launch for consumer products is 2021, with limited distribution to high-end restaurants in 2019. Memphis Meats recently obtained $17 million in Series A funding from investors including Cargill and Bill Gates. The funding will be used to reduce manufacturing costs, quadruple the workforce, and accelerate throughput (Rousseau, O., http://tinyurl.com/GMN-Cargill, 2017).
American food company Hampton Creek, headquartered in San Francisco, has also entered the clean meat scene. According to Eitan Fischer, director of cellular agriculture, the company expects their clean poultry to hit the market in late 2018, at a cost about 30% higher than conventional poultry. The company is building a platform for clean meat production that can be used for multiple species and products. Hampton Creek is in talks to license its clean-meat technology to some of the world’s biggest meat companies, in an effort to radically scale up the technology for cost-efficient production (Rousseau, O., http://tinyurl.com/GMN-hampton, 2017).
FIG. 2. Clean poultry demonstrations from Memphis Meats: A) Southern fried chicken nuggets, and B) Duck à l'orange
Credit: Memphis Meats
The meat of the matter
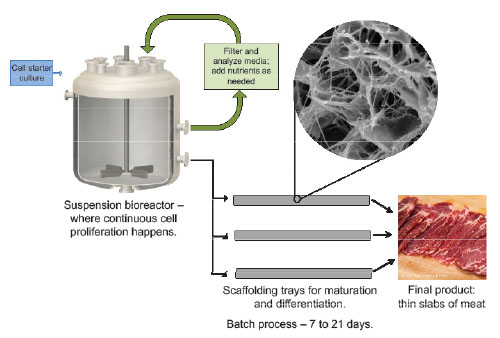
Although each company has its own proprietary techniques for producing clean meat, the overall process is similar (Fig. 3). First, researchers remove starter cells from the animal by a harmless needle biopsy and proliferate them in vitro. The starting cell type could range from embryonic stem cells to fully differentiated muscle cells. Although embryonic stem cells proliferate the most rapidly, they can be difficult to isolate and to direct toward differentiation into a specific cell type. On the other hand, fully developed muscle cells have already differentiated into the desired cell type, but they barely proliferate. Therefore, most companies are using satellite cells—adult stem cells that proliferate at an acceptable rate, but can differentiate only into skeletal muscle cells. Satellite cells, also known as myosatellites, are the cells responsible for muscle regeneration after an injury.
FIG. 3. A conception of how clean meat might be produced at the industrial scale
Credit: The Good Food Institute
After isolating the desired cell type, researchers place the cells in culture medium in a stir-tank bioreactor. The culture medium provides nutrients, salts, pH buffers, and growth factors that allow the cells to proliferate. Historically, fetal bovine serum, which is collected from calf fetuses when a pregnant cow is slaughtered, has been widely used to culture mammalian cells. However, many biomedical companies have developed synthetic or plant-based growth media to avoid ethical issues and problems with batch-to-batch consistency associated with fetal bovine serum. Clean meat companies are working to adapt these serum-free formulations to their specific cell lines.
Satellite cells are anchorage-dependent, meaning that they only grow when adhered to surfaces, and not in suspension. At an industrial scale, cells could be grown in 25,000-L fermenter tanks on microcarrier beads, which would offer a large surface area, or in cell aggregates. Once the satellite cells reach sufficient numbers, researchers alter the culture conditions to cause the cells to differentiate into skeletal muscle cells. The cells fuse into myotubes and begin to express early skeletal muscle markers such as myoD, myogenin, and embryonic isoforms of myosin heavy chain (Post, M. J., http://dx.doi.org/10.1016/j.meatsci.2012.04.008, 2012).
Researchers must apply a mechanical stimulus to trigger muscle protein synthesis and organization into contractile units. This can be accomplished by placing the cells on a collagen-based hydrogel. The muscle cells self-organize into tight fibers within the porous structure of the hydrogel, which creates tension in the fibers. Alternatively, Post’s lab found that by seeding muscle cells around a cylinder of gel, the cells formed a ring-shaped muscle fiber, which could then contract upon itself. Electrical stimulation may further increase muscle protein synthesis, but not enough to justify the energy input required for large-scale production.
For a minced meat product such as hamburger, mature muscle fibers are harvested and assembled into the final product. Structured products, such as a steak or chicken breast, present a much greater technical challenge. For this purpose, cultured meat developers are adopting techniques from the tissue engineering field, which seeks to grow new tissues and organs for medical purposes.
To produce structured meats, satellite cells must be seeded on a three-dimensional scaffold that periodically moves and stretches the developing muscle, simulating an animal’s body. The scaffold, which should be either edible or biodegradable, may consist of a collagen- or cellulose-like gel that is porous to allow the diffusion of medium. To grow large samples, a blood-vessel-like system may be needed to circulate nutrients and oxygen throughout the tissue. 3D printing or spun-fiber platforms could allow tight control over pore size and microstructures within the scaffold (Specht, L., and Lagally, C., http://tinyurl.com/GFI-clean-meat, 2017). Theoretically, fat cells or other cell types could be co-cultured with muscle cells in specific regions of the scaffold to produce, for example, marbling in a steak.
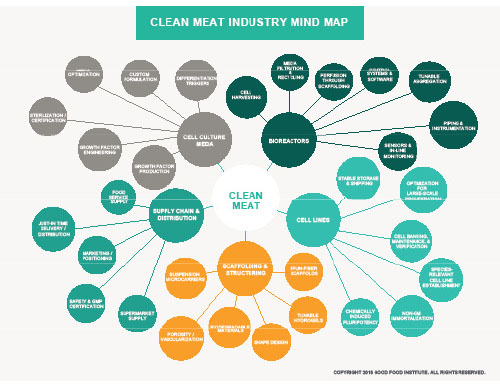
The Good Food Institute, a nonprofit organization that promotes clean meat and plant-based alternatives to animal products, has produced a clean meat industry mind map that illustrates five key areas for advancement of the industry: cell lines, culture media, scaffolding and structuring, bioreactors, and supply chain distribution (Fig. 4).
FIG. 4. A mind map demonstrating the primary elements needed for the large-scale production of clean meat
Credit: The Good Food Institute
Potential benefits of clean meat
Sustainability
Current methods of livestock production are relatively inefficient. The conversion rate of feed to animal protein is about 15% for beef, 30% for pigs, and 60% for chickens (Post, M. J., http://dx.doi.org/10.1111/nyas.12569, 2014). For beef, 1.33 kg of protein (i.e., 7 kg of grain containing 19% protein) must be fed to produce 200 g of protein in meat. In contrast, the scaled-up clean meat procedure requires an estimated 225 g of nutrients (amino acids, glucose, etc.) to produce the same 200 g of protein. Thus, clean meat production is predicted to be about 6 times more efficient than conventional beef production. Because stem cells multiply exponentially, a herd of about 150 cows would be sufficient to feed the entire world, versus the 1.5 billion cows currently on the planet (Post, M. J., http://dx.doi.org/10.1111/nyas.12569, 2014). Fresh samples of stem cells would be periodically removed from live cows and proliferated, since stem cells can become genetically unstable after many divisions. Several companies are exploring options for maintaining genetic stability so that animals can be removed entirely from the process.
However, not everybody is convinced that cattle can be so readily replaced. “Beef cattle play a role in a sustainable food system,” says Daren Williams, director of communications at the National Cattlemen’s Beef Association in Centennial, Colorado, USA. “Cattle consume distillers grains from ethanol production, sugar beet pulp, potato co-products, and plant waste such as carrot tops. If you take cattle out of the equation, you lose the ability to upcycle this plant material into high-quality protein. You also lose the animal byproducts that go into a wide variety of products—industrial, food, and pharmaceutical.”
When a cow is slaughtered for beef, the rest of the carcass does not go to waste. Some of the many byproducts include leather from the hide; soaps, cosmetics, personal care products, and tires from fats; pet food, fertilizer, and gelatin from horns, bones, hooves, and blood; and vaccines and prescription medicines from organs and glands. “At this point, clean meat is still more theoretical than actual,” says Jessica Meisinger, director of scientific education and communication at the National Renderers Association in Alexandria, Virginia, USA. “However, if it became stunningly successful and replaced meat to a sizeable extent, then I imagine there’d be some impact on the volume that renderers receive. And then that would probably have an impact on the price of downstream products such as leather.”
Some companies are developing techniques to produce these downstream products without animals. For example, Geltor (San Leandro, California, USA) has developed a process to produce gelatin with genetically engineered microorganisms. And Modern Meadow (Nutley, New Jersey, USA) is using genetically engineered yeast to produce bovine collagen, which is assembled into fibers and then into sheets of leather that can be tanned, dyed, and finished.
“There are a lot of people looking at ways to provide a sustainable source of protein to feed a growing population,” says Williams. “Meanwhile, back at the ranch, we’re investing in how to produce beef more sustainably.” Williams notes that the National Cattlemen’s Beef Association is conducting a large research project on geographical differences in sustainability. In some cases, land that is not suitable for crop production is fine for grazing cattle. “We could upcycle this land into high-quality protein,” says Williams. “In vitro meat will not provide those benefits. There will likely be negative consequences of taking beef cattle out of the food system.”
Environmental benefits
Clean meat proponents claim that the technology will be much friendlier to the environment than conventional livestock production. Currently, livestock raised for meat consumes about 30% of global ice-free land and 8% of global freshwater, while producing 18% of global greenhouse gas emissions (Tuomisto, H. L., and de Mattos, M. J., http://dx.doi.org/10.1021/es200130u, 2011). In 2011, researchers at the University of Oxford, in the UK, conducted a life cycle assessment (LCA) of the environmental impacts of large-scale cultured meat production. The LCA analyzed the energy, land, and water requirements to produce a minced-meat type product. Because a large-scale production system for clean meat has not yet been reported, the LCA was based on a hypothetical system that used cyanobacteria hydrosylate as the source of nutrients and energy, supplemented with growth factors.
The researchers estimated that the production of 1,000 kg of cultured meat would require 7–45% less energy, 99% less land, and 82–96% less water, depending on the species of meat, compared with the same mass of regular meat produced in Europe (Tuomisto, H. L., and de Mattos, M. J., http://dx.doi.org/10.1021/es200130u, 2011). The greatest savings in energy, land, and water were predicted for cultured beef, and the least for cultured poultry. Cultured poultry actually required more energy to produce than conventional poultry, but less land and water. The researchers predicted that cultured meat would produce 78–96% fewer greenhouse gas emissions than regular meat.
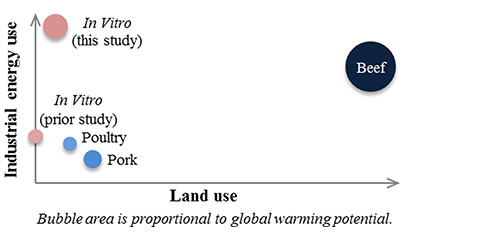
A more recent LCA using different input parameters predicted that clean meat would require less land and water than conventional livestock, at the expense of more energy (Mattick, C. S., et al., http://dx.doi.org/10.1021/acs.est.5b01614, 2015) (Fig. 5). The global warming potential (based on greenhouse gas emissions) was higher for clean meat than for conventional poultry and pork, but lower than for beef. Instead of using cyanobacteria hydrosylate as the culture medium, this analysis used serum-free medium supplemented with soy hydrosylate. Also included in the 2015 LCA, but not in the 2011 analysis, were land, water, and energy requirements for basal medium production, bioreactor cleaning by steam sterilization, a change of medium between the proliferation and differentiation phase, and the production facility.
FIG. 5. An anticipatory LCA predicts that clean meat will require less land but more energy than conventional meat. The analysis also predicts that clean meat will have more global warming potential than conventional poultry and pork, but less than beef. "Prior study" refers to the LCA conducted by Tuomisto and de Mattos in 2011.
Credit: Reprinted with permission from Mattick, C. S., et al., Environ. Sci. Technol. 49: 11941-11949. Copyright 2015 American Chemical Society.
Mattick and coworkers compare the emergence of clean meat to the Industrial Revolution, when automobiles burning fossil fuels replaced work done by horses eating hay (http://dx.doi.org/10.1021/acs.est.5b01614, 2015). Similarly, clean meat production would substitute industrial processes, which require energy inputs, for the internal biological functions performed by animals, such as digestion, respiration, and temperature regulation. “As such, in vitro biomass cultivation could be viewed as a renewed wave of industrialization” with complex trade-offs, the researchers say.
The authors of both LCAs acknowledge that their studies suffer from large uncertainties because no large-scale clean meat facility currently exists. The analyses are conducted for a hypothetical facility using established cell culture techniques, but the technology for producing cultured meat is likely to change rapidly. For example, one of the major energy inputs for the 2015 LCA was steam sterilization of the bioreactor tanks, which is performed in the biopharmaceutical industry. However, this type of sterilization may not be needed for food products—cleaning with soap may be sufficient, or a sterile, biodegradable tank liner may someday be developed for this purpose.
Williams believes that it is too early to say whether clean meat will be more environmentally friendly than traditional livestock production. In contrast to the hypothetical nature of the clean meat LCAs, “we’ve done an LCA on every single input used to produce a pound of conventional beef, down to the HVAC costs and toilet paper used in the meat processing plant,” he says. “We know which areas we can improve.”
Animal welfare
Perhaps the clearest benefit of clean meat would be in the area of animal welfare. No longer would large numbers of animals need to be bred and raised, often in cramped, unsanitary conditions, and then slaughtered by procedures that may or may not be painful. Instead, a small number of companion animals could be kept as living stem cell donors. The People for the Ethical Treatment of Animals (PETA) has supported clean meat, and in 2008 PETA offered a $1 million prize to the first company to bring lab-grown chicken meat to the market by 2012.
Clean meat would allow vegetarians who abstain from meat for ethical reasons to enjoy the flavor and texture of meat without the guilt. However, Post says that vegetarianism is even better for the environment than clean meat. “Cultured meat is not intended for the vegetarian or vegan market. In fact, we would consider it unwanted if vegetarians and vegans start to eat meat as a result of our endeavors,” he says. “That would be the opposite trend of what we are trying to achieve.”
Yet because the majority of people in the world are meat eaters, clean meat has the potential to drastically reduce the suffering of billions of common livestock animals such as cows, pigs, and chickens. In addition, most clean meat platforms are designed to transfer to a variety of species, so less common specialty meats that cause particular animal suffering, such as whale meat or foie gras, could be humanely produced. A Japanese company called Integriculture, Inc., has been culturing chicken pancreas, liver, muscle, and intestinal cells for large-scale clean meat production. They anticipate a commercial launch of “clean foie gras” in 2021 or 2022 (Wan, L., http://tinyurl.com/clean-foie-gras, 2017).
Food safety
Some experts believe that clean meat would be safer for human consumption than traditional meat. Food-borne pathogens such as Salmonella and E. coli are present in the intestinal tracts of livestock and can be transferred to meat during slaughter. Because clean meat is grown in a sterile bioreactor tank, contamination with these and other pathogens can be prevented, or at least readily detected before the product goes to market. Clean meat may also reduce the incidence of emerging diseases such as avian and swine influenza and prion diseases, which have been associated with livestock farming (Post, M. J., http://dx.doi.org/10.1016/j.meatsci.2012.04.008, 2012).
In addition, clean meat might help reduce the antibiotic resistance of pathogens that has resulted from the widespread use of antibiotics in livestock. Mosa Meat and Memphis Meats both claim that they do not require antibiotics because of their sterile lab processes; neither do they need growth-promoting hormones (Zaraska, M., http://tinyurl.com/WP-labmeat, 2016).
Novel foods
By altering cell culture conditions or types of cells, researchers could perhaps produce more healthful meats than those found in nature. Clean meat could be fortified with beneficial fatty acids, such as the omega-3s found in fatty cold-water fish. Saturated fats could be replaced with polyunsaturated fats. Theoretically, cells from different species could be combined to produce meat blends with new flavor, texture, and nutritional profiles.
“Clean meat version 1.0 involves recapitulating as quickly as possible the products that consumers already know and love,” says Liz Specht, senior scientist at the Good Food Institute. “But for version 2.0, the question is, could we actually tailor the composition of these cells to make them, for example, healthier?” She notes that in some cases, simply adding fatty acids or their precursors to the culture medium may effectively seed meat cells with the desired fats. In other cases, researchers might need to genetically modify the metabolic pathways of cells to produce specific fatty acids or other nutrients.
Clean meat challenges
Serum-free cell culture media
Many clean meat companies have prioritized finding synthetic or plant-based substitutes for cell culture media that contains fetal bovine serum, mainly because the serum will become obsolete if clean meat is successful in substantially reducing the slaughter of cattle. Other issues with fetal bovine serum are that its composition is poorly defined, and the quality can be inconsistent. “It can be very hard to get large quantities of high-quality serum,” says Specht. “The other problem is that serum is designed to operate in the context of a living organism, where you need a balance of red lights and green lights to control cell growth. The advantage of serum-free media is that you can only put in the growth factors you want in your particular system, so you’re not hampered by all of the conflicting signals that come with this black-box mixture of growth factors from a calf.”
For many cell types, serum-free culture media that contain recombinant growth factors have already been developed. However, satellite cells require unusually high concentrations of serum for growth, so finding a non-animal substitute that works as well as serum has been challenging (Post, M. J., http://dx.doi.org/10.1111/nyas.12569, 2014). Currently, serum-free media are more costly than media containing fetal bovine serum, which could hamper efforts to scale up clean meat production. In the future, as serum becomes scarcer and recombinant growth factors are mass produced, serum-free media could become the less expensive alternative.
Both Hampton Creek and Memphis Meats claim to have developed viable alternatives to fetal bovine serum, but the companies are tight-lipped about proprietary details. Post says that his group has been reasonably successful in eliminating serum from the culture medium, but they have not yet conducted a high-throughput analysis to optimize a synthetic formulation.
Scaling up and reducing cost
One of the biggest challenges for bringing clean meat to market is transitioning from the lab scale to the industrial scale. Clean meat factories, or “carneries,” would likely resemble beer breweries, with giant tanks for growing meat similar to beer fermentation tanks. One run in a 20,000-L bioreactor would require about 1 month for all steps (cleaning, filling, sterilization, cell proliferation and differentiation, etc.) (van der Weele, C., and Tramper, J., http://dx.doi.org/10.1016/j.tibtech.2014.04.009, 2014). Ten runs per year could supply the meat demand (10 kg per person per year) of about 2,560 people, from a single bioreactor tank. Carneries may partner with existing meat companies that have an established infrastructure for packaging, distributing, and marketing meat.
Post’s 2013 burger was so expensive because the production took place at a laboratory scale (Datar, I, and Luining, D., http://tinyurl.com/NH-cultured-meat, 2015). Through a painstaking process, skilled technicians produced tiny strands of beef in thousands of standard tissue culture flasks, which they then combined to make the burger. Post estimates that scaling up the current technology would reduce the price of the €250,000 ($330,000 USD) burger produced in 2013 to about €8.5 ($10 USD). With technological improvements, clean meat could eventually reach price parity with conventional meat.
“Everyone agrees that the cell culture media will be the main cost driver once clean meat production is at an industrial scale, so I did a pretty detailed cost analysis on the components of the media,” says Specht. “It was surprisingly straightforward to get price parity with conventional meat, without hanging our hat on some kind of technological moonshot.” Simply obtaining bulk pricing for the basal components of synthetic media, such as salts, sugars, amino acids, and lipids, would substantially reduce cost. “Those materials are produced in huge quantities for the food industry, so you can often find metric-ton pricing of those at food-grade quality.”
But the most expensive component of synthetic media is typically the growth factors, which are now produced and used in small quantities for bench and clinical applications. “Growth factors are costing 1 million dollars per gram or something absurd, but it’s a relatively straightforward scaling issue to simply produce those in a recombinant production platform, in the same way that the DuPonts and Novozymes of the world are producing enormous quantities of food processing enzymes,” says Specht.
Structured meats
Thus far, public demonstrations of clean meat have consisted of ground meat, sausages, nuggets, and Spam-like meat. Structured meats such as steaks and chicken breasts will require innovations in scaffolding, vascularization, and other aspects of tissue engineering. Hydrogel scaffolds could be engineered to allow fine-tuning of stiffness, cell adherence, and the controlled release of growth factors (Specht, L., and Lagally, C., http://tinyurl.com/GFI-clean-meat, 2017). In this way, different cell types could be made to differentiate along defined regions, producing, for example, the marbling found in a steak or the flakiness of a fish filet.
According to Specht, clean meat will probably come to the market in phases: first, hybrids of clean meat with plant-based meat; second, ground meat products such as nuggets and burgers; and third, structured meats such as steaks and chicken breasts.
Incorporating fat
Fat imparts flavor, aroma, and texture to meat. So far, clean meat prototypes have lacked fat. Many companies are trying to determine the best way to incorporate fat into clean meat. One option is to culture adipose-tissue-derived stem cells, which differentiate into adipocytes, either together with or separately from satellite cells. An Israeli company called Future Meat Technologies is taking a different approach—using mesenchymal stem cells, which can differentiate into both myocytes and adipocytes, as the starting material (Watson, E., http://tinyurl.com/future-meat, 2017). Mesenchymal stem cells have the added benefit of being able to grow more rapidly, and in less expensive media, than satellite cells.
“Most of the companies are focusing on muscle cells at the moment because it’s much easier to optimize a system when you’ve only got one cell type there,” says Specht. “Also, certain types of intermediate stem cells almost default to adipocytes just by adding certain fatty acids to the growth media, so it’s a bit easier than getting cells to differentiate into muscle.” Specht notes that at the bench scale, researchers have demonstrated co-cultures of skeletal muscle with fat cells. However, companies may find it more efficient to incorporate plant-based fats into clean meat, she says.
Consumer acceptance
Even if the technological hurdles to clean meat commercialization are overcome, there is no guarantee that consumers will accept the products. Similar to genetically modified organisms (GMOs), clean meat may be considered by some to be “unnatural” or a “Frankenfood.” However, cultured meat proponents argue that current meat production systems are far from “natural.”
In 2012, researchers analyzed the reactions and attitudes toward clean meat of 179 meat consumers in Belgium, Portugal, and the United Kingdom (Verbeke, W., et al., http://dx.doi.org/10.1016/j.meatsci.2014.11.013, 2015). Upon learning about clean meat and how it is produced, consumers’ immediate reactions were visceral, including disgust and fear. Many of the study participants viewed clean meat as unnatural and were concerned about the meat’s safety and healthfulness. Although consumers were willing to admit that clean meat could have benefits at the global level (e.g., reducing world hunger or helping starving children in third-world countries), they saw little or no benefit to their own lives. The study participants also worried about the loss of culinary traditions, rural livelihoods, and the preservation of livestock. Some people were concerned that pastures previously occupied by cows would be built upon, swallowing up open spaces and increasing urban sprawl. “For our participants, cultured meat seemed to open a Pandora’s box of unknown societal, environmental, and technological evils,” the researchers say.
A 2016 survey of 673 US consumers revealed that about 65% of respondents would be definitely or probably willing to try clean meat (Wilks, M., and Philips, C. J. C., http://dx.doi.org/10.1371/journal.pone.0171904, 2017). Among the consumers willing to try clean meat, about 33% would be likely to consumer it regularly, and only about 15% would pay more for clean meat than conventional meat.
Although clean meat promises many benefits, the production process must be scaled up and refined to make clean meat marketable. Meanwhile, most ranchers are not exactly concerned about their lifestyle becoming obsolete. “In general, it’s not about us versus them, lab-grown meat replacing traditional meat; it’s about supplying enough protein to feed the world population,” says Williams. “Traditional meat, lab-grown meat, plant-based protein—we’ll need a lot more of everything. But there’s a lot of work to be done to determine the viability and consumer acceptance of in vitro beef.”
Laura Cassiday is an associate editor of Inform at AOCS. She can be contacted at laura.cassiday@aocs.org.
Information
- Benjaminson, M. A., et al. (2002) “In vitro edible muscle protein production system (mpps): stage 1, fish.” Acta Astronaut. 51: 879–889. https://doi.org/10.1016/S0094-5765(02)00033-4
- Datar, I., and Luining, D. (2015) “Mark Post’s Cultured Beef.” New-harvest.org, November 3, 2015. http://tinyurl.com/NH-cultured-meat
- FAO (2009) “The state of food and agriculture. Livestock in the balance.” http://tinyurl.com/FAO-livestock1
- FAO (2011) “World livestock 2011. Livestock in food security.” http://tinyurl.com/FAO-livestock2
- Friedrich, B. (2016) “‘Clean meat’: the ‘clean energy’ of food.” The Good Food Institute blog. http://tinyurl.com/clean-meat
- Hogenboom, M. (2016) “What does a stem cell burger taste like?” BBC News, August 5, 2013. http://tinyurl.com/bbc-meat
- Larsen, C. S. (2003) “Animal source foods and human health during evolution.” J. Nutr. 133: 3893S–3897S
- Mattick, C. S., et al. (2015) “Anticipatory life cycle analysis of in vitro biomass cultivation for cultured meat production in the United States.” Environ. Sci. Technol. 49: 11941–11949. http://dx.doi.org/10.1021/acs.est.5b01614
- Post, M. J. (2012) “Cultured meat from stem cells: challenges and prospects.” Meat Sci. 92: 297–301. http://dx.doi.org/10.1016/j.meatsci.2012.04.008
- Post, M. J. (2014) “An alternative animal protein source: cultured beef.” Ann. N.Y. Acad. Sci. 1328: 29–33. http://dx.doi.org/10.1111/nyas.12569
- Ross, R. (1971) “The smooth muscle cell.” J. Cell Biol. 50: 172–186. http://dx.doi.org/10.1083/jcb.50.1.172
- Rousseau, O. (2017) “Lab-grown meat firms in talks to license tech.” GlobalMeatnews.com, August 10, 2017. http://tinyurl.com/GMN-hampton
- Rousseau, O. (2017) “Cargill invests in lab-grown meat outfit.” GlobalMeatnews.com, August 28, 2017. http://tinyurl.com/GMN-Cargill
- Specht, L., and Lagally, C. (2017) “Mapping emerging industries: opportunities in clean meat.” The Good Food Institute; last updated June 6, 2017. http://tinyurl.com/GFI-clean-meat
- Tuomisto, H. L., and de Mattos, M. J. (2011) “Environmental impacts of cultured meat production.” Environ. Sci. Technol. 45: 6117–6123. http://dx.doi.org/10.1021/es200130u
- Verbeke, W., et al. (2015) “‘Would you eat cultured meat?’: consumers’ reactions and attitude formation in Belgium, Portugal, and the United Kingdom.” Meat Sci. 102: 49–58. http://dx.doi.org/10.1016/j.meatsci.2014.11.013
- Wan, L. (2017) “Lab-made ‘foie gras’: Japan firm claims product could be commercially viable by 2021.” FoodNavigator-asia.com, November 1, 2017. http://tinyurl.com/clean-foie-gras
- Watson, E. (2017) “Future Meat Technologies: the future of clean meat production is local.” FoodNavigator-usa.com, October 3, 2017. http://tinyurl.com/future-meat
- van der Weele, C., and Tramper, J. (2014) “Cultured meat: every village its own factory?” Trends Biotechnol. 32: 294–296. http://dx.doi.org/10.1016/j.tibtech.2014.04.009
- Wilks, M., and Philips, C. J. C. (2017) “Attitudes to in vitro meat: a survey of potential consumers in the United States.” PLoS One 12: e0171904. http://dx.doi.org/10.1371/journal.pone.0171904
- Zaraska, M. (2016) “Lab-grown meat is in your future, and it may be healthier than the real stuff.” The Washington Post, May 2, 2016. http://tinyurl.com/WP-labmeat