Chemically enhanced oil recovery stages a comeback
By Catherine Watkins
November 2009
Nobody knows how much petroleum is left in the ground; estimates range from one trillion to four trillion barrels. Current global production of petroleum is about 85 million barrels/day (bbl/d), or more than 30 billion bbl/year. OPEC (the Organization of the Petroleum Exporting Countries) expects demand by developing countries to increase global production to 107 million bbl/d by 2030.
Here is one more statistic that signals unparalleled opportunity for surfactants researchers and manufacturers: Only about 30% of available oil and gas has been extracted from most existing wells. That leaves chemically enhanced oil recovery (CEOR) on the short list of methods available to boost oil production in the secondary and tertiary phases of oil recovery (see sidebar, page 684).
Some industry observers are leery of such talk. They remember the research boom in the 1970s and 1980s when the oil crisis pushed up the price of oil and research flourished. They also remember the bust that occurred once oil prices fell. CEOR in the 21st century, however, is significantly different: Custom-made surfactants used at much lower concentrations in tandem with new technology have dramatically changed the outlook.
"Shell is quite bullish on surfactant-based CEOR," Kirk Raney, a senior staff research engineer at Shell International Exploration and Production Inc. in Houston, Texas, USA, explained. "We feel CEOR is a large prize that will maximize our existing resources and will be an important component of meeting the world's energy challenge."
Shell is not alone in that belief. Celanese, Rhodia, Tiorco (Stepan's joint venture with Nalco), Sasol, Oil Chem Technologies, and Surtek are just a few of the participants in the CEOR market anticipating future growth. All of them are bullish about CEOR and happy to explain why.
ECONOMICS
The development that most affects the economics of CEOR in the 21st century is the change in the concentration of surfactants used in reservoir flooding. CEOR in the 1970s and 1980s focused on "micellar" flooding with 2-12% surfactant. Now, the surfactant concentrations "are in the range of only 0.1-0.5%," said Kon Wyatt, vice president of engineering at Surtek Inc. in Golden, Colorado, USA. As a full-service firm, Surtek has provided engineering, laboratory, and operational expertise since 1978 to oil companies seeking to improve oil recovery.
"The bottom line is that the new surfactant formulations cost in the range of $0.90-$2.75/bbl injected, where a micellar formulation would cost $20-$75/bbl injected at today's chemical costs," Wyatt noted in a recent article in The American Oil & Gas Reporter (52:102-105, 2009). "Establishing a successful CEOR operation is a long-term commitment, but the rewards can be substantial and long-lived, even at oil prices as low as $20-$25/bbl."
FIRST WAVE OF RESEARCH
One longtime CEOR researcher who was involved in the first wave of CEOR research is Gary Pope, a professor in the Department of Petroleum and Geosystems Engineering and director of the Center for Petroleum and Geosystems Engineering at The University of Texas at Austin (UT; USA). Pope, who once worked at Shell, counts among his staff AOCS member and Samuel Rosen Memorial Award-winner Upali Weerasooriya, who is director of surfactant development for the center.
Pope points first to the surfactants used in the 1970s, when petroleum sulfonates were the surfactants of choice. "Now we know what the molecular structures are and how to vary them," he said. "Those structures can be made at about the same cost, or even less, than before, mostly at under $2/pound," he added.
Then there are the differences in the scope and speed of research. In the old days of CEOR, petroleum companies would put 20 staff members on a pilot study for two years. "At today's cost that would be about $20 million in labor alone at $500,000/person/year," Pope noted. Now, with high-throughput methods and a willing graduate student, the same project can be completed in about three months, he said.
The UT Center currently has separate funding for 16 projects, including surfactant selection for CEOR at Cairn India's large oilfield in Rajasthan. In addition, the group has worked with many companies all over the world. Interest in collaborating with the Center has accelerated in the last six months, Pope reported. "Once the price of oil hit $50 again [after a low of around $35 in March 2009], interest just exploded," he said.
Further changes differentiating CEOR efforts in the 1970s and 1980s from the present, according to Pope and Weerasooriya, include advances in polymer science as well as in oil drilling technology, with horizontal well drilling, advanced reservoir characterization, and 3D seismic exploration now available in the oil recovery toolkit.
Pope and Weerasooriya also point to breakthroughs in the UT laboratory within the past year. "We now have an alkaline that will work in hard brine," they said, "allowing us to conduct alkaline-surfactant-polymer flooding without softening the water. We also have large branched surfactants available at relatively low cost. In addition, we have developed a process to use sulfates, as opposed to sulfonates, that can be employed at high temperatures."
The UT scientists are working with Shell, BASF, Harcros Chemicals, Huntsman, Stepan, and others to commercialize the many products developed in their laboratory-which points to another difference between the CEOR boom of the 20th century and the current boom: "During the first boom," Pope said, "there was a complete disconnect between oil companies and chemical companies, even when it was the same company."
OTHER NEW WORK
Another prominent CEOR researcher is AOCS member and Samuel Rosen Memorial Award-winner Paul Berger, who is vice president and technical director of Oil Chem Technologies, Inc., in Sugar Land, Texas, USA. The company recently was listed as No. 2 on Inc. magazine's 2009 list of fastest-growing companies in the energy sector, up from No. 6 the previous year.
Oil Chem has been working since 1995 on CEOR and currently has 20 active projects with an annual recovery of up to one million barrels of oil. To date, the company has successfully injected more than 50 million pounds of surfactants at oilfield projects all over the world. In a recently completed Canadian CEOR project, the oilfield went from producing 300 bbl/d to producing over 2,000 bbl/d, an increase of more than 600%.
"We are starting to look at very high-molecular-weight glycerides (C18-C24) from vegetable oils such as rapeseed, sunflower, safflower, meadowfoam, jatropha, and another I can't talk about just yet," said Berger. "They can be used to make viscoelastic surfactants that can give you viscosity in reservoirs with high salinity and temperatures where polymers won't work," he added. "Viscoelastic surfactants actually seek out the oil rather than staying in solution in the water-we call them ‘smart surfactants.'" They are also used to make novel anionic ether sulfonates that are resistant to high salinities and temperatures.
Oil Chem's viscoelastic surfactants (VS) form viscoelastic solutions in injection fluids at 0.1-1.0 wt%, reducing interfacial tension to below 0.01 millinewtons/meter. They divert injection fluids to oil-bearing sites by reducing viscosities as they contact oil, allowing injection fluids to flow into oil-bearing portions of the reservoir, Berger said. They also exhibit high thermal stability, he noted. They are particularly useful when used alone in tight formations with high temperatures and salinities. Used with polymers, VS can provide some residual viscosity when contacting oil to help the oil sweep out of microscopic pores in the reservoir while maintaining high viscosity in the water zones. Unlike polymers, VS are not permanently degraded by high shear, Berger noted. "Although shear converts their worm-like micelles to a spherical shape, this process is reversible. They become less viscous under shear but their viscosity returns when the shear is removed."
Oil Chem Technologies is developing five major lines of VS for use depending on the type of oil, pH, water hardness, and temperature in the reservoir system. Berger estimates that the company's present line of surfactants is used in roughly 80% of current CEOR projects worldwide. The company has also developed surfactants for recovering oil from tar sands, heavy oil deposits, shale, and for reservoirs having high concentrations of total dissolved solids and high temperatures.
In other work, Sasol North America Inc. is investigating microemulsions that can be applied to CEOR. Research led by AOCS member Charles Hammond, a research associate based in Westlake, Louisiana, USA, involves adjusting the optimum salinity of anionic microemulsions with alcohol ethoxylates. His presentation at the 2009 AOCS Annual Meeting & Expo illustrated that the optimum salinity of alcohol propoxylate sulfates can be shifted by the addition of alcohol ethoxylates and influenced by the degree of alcohol ethoxylation, the concentration of alcohol ethoxylate, and the structure of the alkyl group.
"With the ever-growing need for energy, and the high volume of hard-to-obtain oil underground, CEOR is inevitable," Hammond said. "Although there are several tertiary methods available to pursue oil recovery, we believe that there is great potential for surfactants. In fact, Sasol Olefins & Surfactants is working on generating fundamental surfactant structure-property knowledge that will be needed by both surfactant manufacturers and formulators for CEOR as this market continues to grow."
MARKET POTENTIAL
The market potential for surfactant use in CEOR is substantial. One analysis by Shell's Kirk Raney suggests that annual demand for surfactants for CEOR (C14-C28) could eventually be as high as 12.5 million metric tons (MMT)/year. That figure is roughly double the existing total surfactant market demand of around 6 MMT/year (predominantly C10-C16). [See Fig. 1 for a production route flowchart.
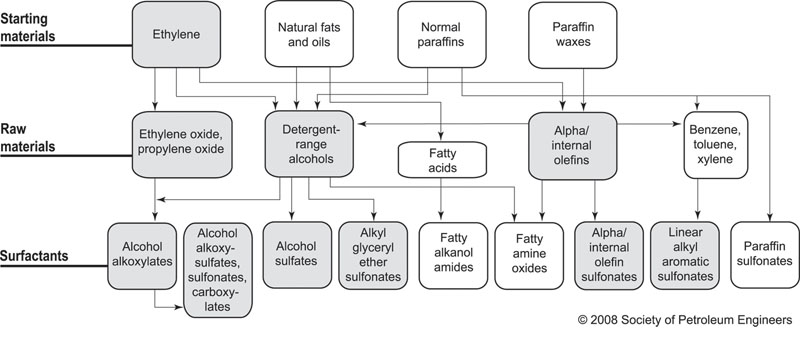
FIG. 1. Different routes to surfactants. Source: Barnes, J.R., J.P. Smit, J.R. Smit, P.G. Shpakoff, K.H. Raney, and M.C. Puerto, Development of Surfactants for Chemical Flooding at Difficult Reservoir Conditions, Paper SPE-113313-MS, presented at the Society of Petroleum Engineers/Department of Energy Improved Oil Recovery Symposium, Tulsa, Oklahoma, USA, April 19-23, 2008. Shaded boxes: Shell Chemical commercial routes and surfactants
"Assuming oil prices stay at least at current levels, several large surfactant floods are scheduled by the oil industry to occur in the 2015-2020 time frame," Raney said. "Each could consume on the order of 25,000-50,000 metric tons of surfactant per year at full implementation. This projected level of demand would make a significant impact on the synthetic surfactant market. However, it is important to note that the surfactant hydrophobes of most interest to CEOR researchers today are different than those currently produced at large scale in the detergent industry," he added. "Therefore, if current capacity is used to manufacture surfactants for CEOR, changes to the process will be needed."
But are the major petroleum companies ready to gamble again on CEOR?
"There is a tremendous amount of interest by oil companies in doing the groundwork for CEOR so that when the price of oil increases, they are ready," said Tom Waldman, director of sales and marketing for Tiorco of Denver, Colorado, USA, which provides custom-engineered chemical solutions for EOR. Tiorco is a joint venture formed in September 2008 by Stepan Co., a surfactant manufacturer located in Northfield, Illinois, USA, and Nalco, a water treatment company based in Naperville, Illinois.
There is good reason for oil companies to be cautious about CEOR. "The return on investment is slow and it is a huge financial risk," noted Bob Krueger, global business director, oilfield, for Stepan. "A company can spend $50 million on flood-pumping equipment and chemicals and not see the first return on investment for 24 months. By comparison, it takes about five days to drill 10,000 feet," he added.
Nonetheless, with new, more efficient molecules that make more economic sense than before, CEOR clearly will-slowly but surely-alter the surfactants industry.
"Although it has taken years, we have sold the oil companies on the idea that using chemical energy in place of some mechanical energy can have a net-net positive effect," Krueger concluded.
Catherine Watkins is associate editor of inform and can be reached at cwatkins@aocs.org
Oil production primer
Oil recovery occurs in three phases: primary, secondary, and tertiary. But because each reservoir has its own unique geologic and chemical makeup, the lines separating the three phases often are blurred.
Primary recovery occurs when internal pressure in a reservoir is high and oil all but spurts out of the ground. About 10% of available oil typically is removed in this phase. Secondary recovery, which can remove an additional 20-40% according to the US Department of Energy, consists of repressurizing the reservoir by pumping water or gases into it. The tertiary phase involves enhanced oil recovery (EOR) techniques that modify the reservoir system to allow the oil to flow more freely out of the well.
EOR techniques-which sometimes are also required in the primary and secondary phases of recovery-fall into three main categories: thermal, chemical, and solvent displacement. All of these involve the injection of fluids into the reservoir to generate fluid properties or interfacial conditions that are more favorable for oil displacement. The injection of steam heats the oil and makes it more fluid. Gas injection (generally natural gas, nitrogen, or carbon dioxide) pushes additional oil to a production well. Other gases that dissolve in the oil can be used, thereby thinning it and improving the flow rate.
Chemical EOR generally involves flooding a reservoir with an alkaline-surfactant-polymer combination, a surfactant-polymer mix, or a polymer-only injection. Current thinking suggests that the alkali reacts with acids and esters in the crude oil to form surfactants "that combine with the injected surfactant to produce synergistic mixtures at the oil/brine interface," according to Paul Berger and Christie Lee of Oil Chem Technologies, Inc., in Sugar Land, Texas, USA. "The alkali is also claimed to reduce the amount of surfactant adsorption onto the formation, especially in limestone reservoirs," they continue. In addition, the surfactant reduces the interfacial tension and the polymer reduces viscosity, thereby increasing flow.
Polymers used in EOR include polyacrylamide, AMPS (2-acrylamido-2-methyl propanesulfonic acid) copolymer, xanthan gum, and scleroglucan. To date, anionic surfactants have performed best in EOR. In general, highly branched hydrophobes are needed for low-viscosity micelles and microemulsions. Although ethylene has been the feedstock of choice in the past, Berger and Lee have found that naturally derived surfactants with a double bond serve equally well as branched petroleum-derived products. "Our patent US 7,556,098 illustrates this," Berger noted. Researchers are also looking at using genetically modified microorganisms that can produce biosurfactants in situ.
The decision about which surfactant to use depends on factors such as reservoir temperature, pressure, depth, salinity, and permeability. Each oilfield is different and has to be characterized before the most effective surfactant can be modeled and produced. The advent of high-throughput experimentation has allowed researchers to custom-make surfactants of high purity and specificity at relatively low cost.
information
Two presentations from the 2009 AOCS Annual Meeting & Expo mentioned in this article are available online to AOCS Surfactants and Detergents (S&D) Division members. They are Paul Berger and Christie Lee's talk on viscoelastic surfactants and Charles Hammond and Geoff Russell's presentation on adjusting optimum salinity of anionic microemulsions used in enhanced oil recovery. The slides are available at www.aocs.org/member/division/surfact/ . To join the S&D Division or for help accessing the papers, contact Doreen Berning at doreenb@aocs.org or +1-217-693-4813.
A presentation by Gary Pope of the University of Texas at Austin entitled "Overview of Chemical Flooding" can be found at http://eori.gg.uwyo.edu/downloads/Overview of chemical EOR.pdf.
Barnes, J.R., J.P. Smit, J.R. Smit, P.G. Shpakoff, K.H. Raney, and M.C. Puerto, Development of Surfactants for Chemical Flooding at Difficult Reservoir Conditions, Paper SPE (Society of Petroleum Engineers)-113313-MS, presented at the SPE/Department of Energy Improved Oil Recovery Symposium, Tulsa, Oklahoma, USA, on April 19-23, 2008.
Levitt, D.B., A.C. Jackson, C. Heinson, L.N. Britton, T. Malik, V. Dwarakanath, and G.A. Pope, Identification and Evaluation of High-Performance EOR Surfactants, SPE Reservoir Evaluation & Engineering 12:243-253, 2009.
