Understanding pulse anti-nutrients
By Mark Messina
January 2020
- Pulses have desirable health and nutritional attributes, but raw pulses contain relatively high levels of antinutrients.
- These compounds may reduce protein digestibility and the absorption of minerals or even have direct detrimental effects.
- Soaking and heating eliminates, reduces, or inactivates many of them.
- The health advantages of pulses greatly outweigh any residual antinutrient content of properly processed pulses.
Pulses have desirable health and nutritional attributes and their consumption may help mitigate climate change [1,2] , but they are an underutilized source of nutrition in most developed countries [3–5] despite recommendations by health agencies to increase their intake [6]. This could be due in part to concerns about antinutrients [7].
Antinutrients are classically defined as compounds that inhibit nutrient use, but the broader definition used in this article includes compounds that have direct detrimental effects independent of nutrient use [8]. Many plant foods contain antinutrients. For example, cruciferous vegetables contain glucosinolates, which can inhibit thyroid function [9]. Potatoes and tomatoes contain solanine, a glycoalkaloid poison that interacts with mitochondrial membranes [10]. These types of compounds are ubiquitous throughout the plant kingdom and often serve as defense molecules or phytoalexins that enhance plant survival by warding off predators, and their concentration increases when plants are stressed [11]. Antinutrients are not limited to plants; raw eggs contain avidin, which inhibits biotin absorption [12].
Such foods can be safely consumed, because conventional food processing (soaking, heating, milling, germination, and fermentation) greatly reduces their concentrations and/or because only consuming excessive amounts is harmful. Many antinutrients in plants are also under study for their potential health benefits. For example, glucosinates are widely touted for their chemopreventive properties [13]. The isoflavones in soybeans are purported to exert several benefits in postmenopausal women, such as improving bone mineral density [14] and alleviating hot flashes [15], and some antinutrients discussed in this article have proposed benefits as well [16–20].
The antinutrients in pulses include protease inhibitors, phytate, oxalate, lectins, tannins, saponins, polyphenols, amylase inhibitors, and oligosaccharides. This article focuses only on the first four and the two processes that are most applicable to pulses: soaking and heating. Also, while the soybean is not considered a pulse (pulses are the dried edible seeds of plants in the legume family, and soybeans are too high in fat to be dry), it has been extensively researched and is often used as a comparator.
Protease inhibitors
Proteolytic enzymes, or proteases, catalyze the hydrolytic cleavage of specific peptide bonds in their target proteins. They are widespread in the plant kingdom and form less active or fully inactive complexes with their cognate enzymes. Protease inhibitors are induced in response to injury or attack by insects or pathogens [21]. Protease inhibitors common in pulses include trypsin inhibitors, the Kunitz Trypsin Inhibitor, and the Bowman-Birk Inhibitor [22]. Protease inhibitors can comprise as much as 6% of a legume’s total protein content [23,24].
The extent to which their activity is destroyed by heat is a function of temperature, heating duration, particle size, and moisture conditions. Relatively little protease inhibitor remains in properly processed pulses. Soaking and cooking different beans at 95°C in water (1:5 seed:water) in a beaker for 1 h destroys 100% of the chymotrypsin inhibitor activity and 80 to 100% of the trypsin inhibitor activity [25], and a similar process [26] was shown to eliminate 100% of the trypsin inhibitor activity of Canadian and Egyptian cowpea, kidney beans, and pea.
Phytate
Phytate, or inositol hexaphosphate (IP6), is a naturally occurring compound found in whole grains and legumes. It is the major storage form of phosphorous, comprising 1–5 % by weight in cereals, legumes, oil seeds, and nuts [27]. It represents 50–85 % of the total phosphorous in plants [28], and grains are the primary sources of phytate in Western diets [29].
Phytic acid is made up of an inositol ring with 6 phosphate ester groups. It is the most abundant form of myo-inositol phosphate found in mature, unprocessed plant-based foods [20]. Phytic acid chelates cations, forming insoluble complexes with minerals in the upper gastrointestinal tract [31]. These complexes cannot be digested or absorbed by humans because of the absence of intestinal phytase [43,44]. However, during some food-processing and storage practices, IP6 is dephosphorylated to lower myo-inositol phosphate forms, some of which no longer inhibit mineral absorption [16,30]. However, most of the phytate in beans is present as IP6 or IP5 [32].
While moist heat at high temperature destroys most protease inhibitor activity, boiling beans only reduced phytate content by an average of about 20% [32,33]. The reduction varies markedly among species. Boiling reduced about 50% of the phytate content from cowpea, pea, and kidney beans [26], and most of the loss was from soaking prior to heating.
Compared to heme iron, the non-heme iron found in plant foods is poorly absorbed due to iron absorption inhibitors, mostly phytate. Consequently, the Institute of Medicine of the National Academies set the iron recommended daily allowance (RDA) for vegetarians, who consume only nonheme iron, at 1.8 times the non-vegetarian RDA [34], and the European Food Safety Authority set zinc requirements based on four different levels of phytate intake (300, 600, 900, and 1200 mg/d) [35].
Iron absorption from single meals based on black beans, lentils, mung beans, and split beans was found to be very low, ranging from only 0.8% to 1.9% [36]. However, since multiple plant components affect iron absorption, the contribution of phytate to the poor iron absorption of beans could not be determined. On the other hand, genetically reducing the phytate content of common beans by 90% leads to an approximate doubling of iron absorption [37].
Phytate also inhibits calcium absorption from legumes [38,39]. The fractional calcium absorption from milk and high and low-phytate soybeans was found to be 0.377, 0.310, and 0.414, respectively [38]. Zinc intake is also inhibited by phytate but to a lesser extent than iron [40].
Since most of the information about the impact of inhibitors on mineral absorption comes from single meal studies, Cook, et al. [41], concluded that “in the context of a varied Western diet, nonheme-iron bioavailability is less important than absorption studies with single meals would suggest.” This may also be true for zinc [42,43].
Importantly, in contrast to an initial report by Brune, et al. [44], Armah, et al. [45], recently found that habitual consumption of a high-phytate diet minimizes its inhibitory effects. Differences in study design could explain the discrepancy: The average daily phytate intake in the more recent study was 1190 mg, whereas the average intake in the initial work was only 323 per day, which is quite low. Whether this adaptation applies to other minerals inhibited by phytate has not been established. Several researchers have argued that iron absorption from soybeans is greater than the data suggest because the traditional methodology used to assess iron absorption is not appropriate for measuring the bioavailability of iron in the form of ferritin, a form of iron that may be insensitive to iron inhibitors [46–48]. The percentage of iron in the form of ferritin ranged from 10 to 41%.[49] among six legumes, but it is premature to reach any definitive conclusions about iron ferritin bioavailability. It is also worth noting that vegetarian iron stores are lower than iron stores of non-vegetarians, although the clinical significance of these lower stores, if any, is a matter of debate [63,64].
Oxalate
Oxalic acid and its salts occur as end products of metabolism in several plant tissues. Oxalic acid forms water-soluble salts with Na+, K+, NH4+ ions. It also binds with Ca2+, Fe2+, and Mg2+, rendering these minerals unavailable for absorption. However, Zn2+ appears to be relatively unaffected [50], and although there is some evidence that oxalate inhibits iron absorption [51], not all data indicate this is the case [52]. The primary concern from a nutrient standpoint is oxalate’s impact on calcium absorption. Also, a high-fiber and high-oxalate diet worsens calcium balance compared to a high-fiber or high-oxalate diet alone [53].
It is well known that calcium absorption of high-oxalate vegetables is extremely poor. The absorption index (calcium absorption of test source divided by the milk calcium absorption value) of the high-oxalate vegetables Chinese spinach, rhubarb, and sweet potatoes was 0.257, 0.235, and 0.423, respectively, whereas from the low-oxalate vegetables Chinese mustard greens and Chinese cabbage flower leaves, it was 1.080 and 1.097, respectively [54].
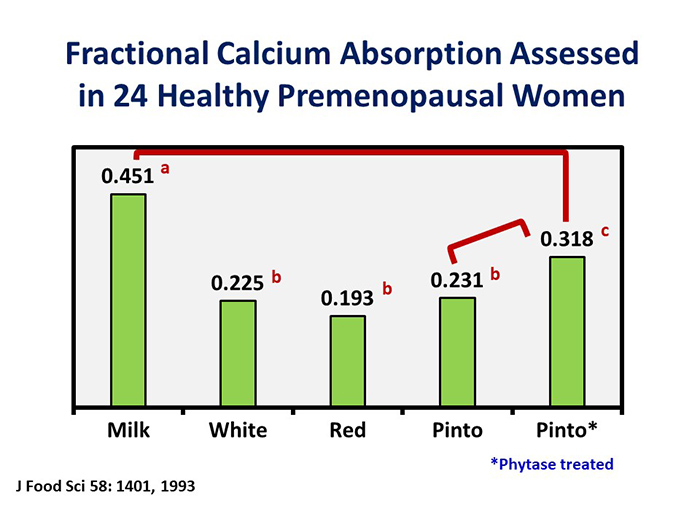
The authors of this study concluded that the likely explanation for the phytase-resistant calcium absorbability deficit of pinto beans—fractional calcium absorption from pinto beans to which phytase was added was still lower than from milk (Fig. 1)—was due to the oxalate content. Interestingly, despite its high oxalate and phytate content, the absorption of calcium from soybeans [38] and calcium-fortified soymilk [55] and calcium-set tofu [56] is comparable to or only slightly lower than from cow’s milk.
FIG. 1.
The risk of developing kidney stones is an additional concern with high-oxalate foods. The incidence and prevalence of kidney stones is increasing in much of the world [57]. In the United States, the prevalence of kidney stones increased from 3.2% in 1976 to 1980 to 5.2% in 1988 to 1994 [58]. According to the most recent estimate from the National Health and Nutrition Examination Survey (2007–2010), the prevalence of kidney stones is 8.8% (10.6% among men, 7.6% among women) [59].
Approximately 80% of the kidney stones in industrialized countries are composed of calcium salts—usually calcium oxalate and, to a lesser extent, calcium phosphate [60]. Urinary oxalate is the single strongest chemical promoter of kidney stone formation, as it has a much bigger effect on stone formation than urinary calcium [61]. As much as 50% of the oxalate in the urine comes from food when a typical diet containing 10–250 mg dietary oxalate is consumed; the other half comes from endogenous synthesis [77–78].
The American Dietetic Association classifies foods containing >10 mg oxalate per serving to be high-oxalate foods and recommends that kidney stone formers limit their intake. Further, it is recommended, especially for individuals who are hyperabsorbers of oxalate, to limit total oxalate intake to no more than 100 mg per day [79,80]. In fact, Han, et al. [62], recently recommended consuming no more than 40–50 mg oxalate per day to prevent recurrence of kidney stones. Several studies indicate that stone formers on average may absorb about 50% more oxalate than healthy individuals [82,83].
Compared to high-oxalate foods such as Red Swiss chard, spinach, and rhubarb, which contain about 1,000 mg per 100 g wet weight [63], the oxalate content of soaked and cooked legumes is low, ranging from about 60 to 180 mg/100 g dry weight (approximately 20 to 60 mg wet weight) [33]. Consuming many pulses would still not be recommended for those at risk of developing or who have a history of kidney stones. However, Chai, et al. [63], reported that cooked garbanzo beans, mung beans, lentils, large lima beans, green split peas, yellow split peas and blackeye peas each contained no more than 10 mg per 100 g wet weight. So, these foods would be acceptable.
Boiling vegetables decreased total oxalate content 30% to 87%, primarily by loss of soluble oxalate (Table 1). In spinach and carrots, the decrease in total oxalate corresponded to the amount of oxalate found in the cooking water [64]. Similar results were found when New Zealand and Thai vegetables were boiled [65,66,67], so boiling vegetables may be an option to reduce soluble oxalate, if the cooking water is not consumed. In contrast, baking potatoes [64] or roasting peanuts [67] or sesame seeds [68] does not reduce oxalate. For many legumes, cooking reduced oxalate content by 31 to 66%, most of which occurred as a result of soaking [33].
TABLE 1. Oxalate content (mg/100 g wet weight) of cooked legumes (boiled and drained)
The relative amount of soluble and insoluble oxalate is an important consideration. Insoluble oxalate is bound to another molecule such as calcium or magnesium, which makes it difficult to absorb. Taking calcium supplements with a meal to bind oxalate and prevent its absorption is sometimes recommended for kidney stone patients [69]. Soluble oxalate is not attached to another molecule, so is more easily absorbed. The relative amount of each form in foods varies markedly. For example, the ratio of soluble to insoluble oxalate in wheat bran, oat bran, barley bran, red kidney beans, and white beans was 0.38, 0.99, 0.81, 0.21, and 0, respectively [70].
Since solubility affects absorption, foods with higher amounts of soluble oxalate lead to higher urinary oxalate levels. To illustrate, cinnamon (3.5 g) and turmeric (3.2 g) each provide 63 mg oxalate, but approximately 91% of the oxalate in turmeric and 6% in cinnamon is soluble. Consequently, turmeric consumption significantly increases urinary oxalate levels in patients with a history of kidney stones, whereas cinnamon has no effect [70]. Similarly, 37 mg of the 120 mg oxalate in almonds was determined to be soluble, whereas only 6 of 100 mg in black beans was. Among 6 male and 5 female study participants, 5.9% and 1.8% of the oxalate in almonds and black beans was absorbed, respectively [71]. Not surprisingly, the authors concluded that for individuals with hyperoxaluria or who are prone to forming oxalate-containing kidney stones, eating almonds was riskier than eating beans.
Many factors besides oxalate intake affect kidney stone formation. Increasing water intake, decreasing animal protein, limiting sodium, and increasing fruit and vegetable consumption are often recommended for prevention [72]. These other risk factors may account for why a preliminary British survey found that vegetarians were only half as likely to develop kidney stones as non-vegetarians [73].
Recent data also show that adhering to the DASH (Dietary Approaches to Stop Hypertension) diet is very protective against renal stones [74]. Interestingly, phytate strongly inhibits calcium oxalate crystal formation in vitro [75]. This may be why an analysis from the Nurses' Health Study II, which included 96,245 female participants, found that over an 8-year follow up period, phytate intake was associated with a 63% reduced risk of stone formation [76].
Lectins
Beans are the main source of dietary lectins [77], but these carbohydrate-binding proteins are widely distributed throughout the plant kingdom [78]. More than 500 lectins have been identified [79]. The lectin ricin was isolated from castor beans (Ricinus communis L.) in the late 1800s [80]. The lectin in soybeans was discovered in the 1950s [81].
In plants, lectins function as nitrogen storage compounds but also have a defensive role, protecting plants against pests and predators [82,83] and possibly mediating specificity in Rhizobium-legume symbiosis [84]. Lectins are capable of specific recognition of and binding to carbohydrate ligands.
The term “lectin” is broadly used to denote “all plant proteins possessing at least one non-catalytic domain, which binds reversibly to a specific mono- or oligosaccharide. [83]”
Undigested plant lectins remaining in the gut may bind to a wide variety of cell membranes and glycoconjugates of the intestinal and colonic mucosa, leading to deleterious effects on the mucosa as well as on the intestinal bacterial flora and other inner organs [80,85]. The severity of these adverse effects may depend upon the gut region to which the lectin binds [86].
High resistance to proteolytic degradation results in nutritionally significant amounts of intact and highly reactive forms of certain dietary lectins within the gut lumen [87]. Intact peanut agglutinin (PNA) can be detected at up to 5 mg/ml in the systemic circulation after eating peanuts, enough to stimulate proliferation of epithelial cells [88]. Detection with a heme agglutination assay implies retained biological activity.
Several cases and outbreaks of lectin poisoning from consumption of raw or improperly processed kidney beans have been reported [89–93]. A dose of purified ricin powder the size of a few grains of table salt can kill an adult human [94]. Because lectin proteins bind to cells for long periods of time, they can potentially cause an autoimmune response and are theorized to play a role in inflammatory conditions like rheumatoid arthritis and type 1 diabetes [87,95].
Lectins also bind to carbohydrates in ingested food, and limit or change their potential hydrolysis and absorption from the gut. Thus, they reduce the amount of energy for growth and maintenance [96,97]. They can also increase proliferation of pathogenic bacteria in the gut [98] and increase intestinal permeability [99].
There is a large variation in the lectin content of different varieties of raw legumes [100,101]. The lectin content of soybean varieties varies as much as fivefold [102,103]. The most practical, effective, and commonly used method to abolish lectin activity is aqueous heat treatment. The lectin activity in fully hydrated soybeans, kidney beans, faba beans, and lupin seeds was completely eliminated under conditions where the seeds are first fully soaked in water and then heated in water at or close to 100°C [104,105]. A more recent study showed that soaking and cooking beans eliminated most, but not all, of the lectin activity [33]. Boiling at 95°C for 1 hour eliminated ~97% of the activity in different varieties of peas and lentils, >99% in different varieties of beans, 99% in two varieties of fava bean, and >99% in soybeans.
Given that exposure to lectins is widespread [106], an editorial in the British Journal of Medicine 20 years ago asked, “Why don't we all get insulin-dependent diabetes, rheumatoid arthritis, IgA neph¬ropathy, and peptic ulcers [95]?” The answer was, “partly because of biological variation in the glycoconjugates that coat our cells and partly because these are protected behind a fine screen of sialic acid molecules, attached to the glycoprotein tips. [107]” Clinical evidence that any residual lectin activity in properly prepared pulses (soaking and boiling) leads to detrimental effects is lacking.
Mark Messina, PhD, is the executive director of the Soy Nutrition Institute, president of Nutrition Matters, Inc., a nutrition consulting company, and is an adjunct associate professor at Loma Linda University (California, USA). He has spent the past 30 years focusing primarily on the health effects of soybeans and consumes legumes daily. He can be contacted at markjohnmessina@gmail.com.
References
- Foyer, C.H., H.M. Lam, and H.T. Nguyen, et al., Neglecting legumes has compromised human health and sustainable food production, Nature plants 2: 112, 2016.
- Polak, R., E.M. Phillips, and A. Campbell, Legumes: health benefits and culinary approaches to increase intake, Clin. Diabetes. 33: 198–205, 2015.
- Gardner, C.D., J.C. Hartle, and R.D. Garrett, et al., Maximizing the intersection of human health and the health of the environment with regard to the amount and type of protein produced and consumed in the United States, Nutr. Rev. 77: 197–215, 2019.
- Miller, V., A. Mente, and M. Dehghan, et al., Fruit, vegetable, and legume intake, and cardiovascular disease and deaths in 18 countries (PURE): a prospective cohort study, Lancet 390: 2037–49, 2017.
- Shan, Z., C.D. Rehm, and G. Rogers, et al., Trends in dietary carbohydrate, protein, and fat intake and diet quality among US adults, 1999-2016, JAMA. 322: 1178–87, 2019.
- Herforth, A., M. Arimond, and C. Alvarez-Sanchez, et al., A global review of food-based dietary guidelines, Adv. Nutr. 10: 590–605, 2019.
- Bresciani, A. and A. Marti, Using pulses in baked products: lights, shadows, and potential solutions, Foods 8, 2019.
- Essers, A.J., G.M. Alink, and G.J. Speijers, et al., Food plant toxicants and safety risk assessment and regulation of inherent toxicants in plant foods, Environ. Toxicol. Pharmacol. 5: 155–72, 1998.Essers AJ, Alink GM, Speijers GJ, et al. Food plant toxicants and safety Risk assessment and regulation of inherent toxicants in plant foods. Environ Toxicol Pharmacol. 1998;5(3):155-72.
- Heaney, R.K. and G.R. Fenwick, Natural toxins and protective factors in Brassica species, including rapeseed, Nat. Toxins. 3: 233–7; discussion 42, 1995.
- Barceloux, D.G., Potatoes, tomatoes, and solanine toxicity (Solanum tuberosum L., Solanum lycopersicum L.), Dis. Mon. 55: 391–402, 2009.
- Rousseau, S., C. Kyomugasho, and M. Celus, et al., Barriers impairing mineral bioaccessibility and bioavailability in plant-based foods and the perspectives for food processing, Crit. Rev. Food Sci. Nutr. 1–18, 2019.
- Sweetman, L., L. Surh, and H. Baker, et al., Clinical and metabolic abnormalities in a boy with dietary deficiency of biotin, Pediatrics 68: 553–8, 1981.
- Dinkova-Kostova, A.T. and R.V. Kostov, Glucosinolates and isothiocyanates in health and disease, Trends Mol. Med. 18: 337–47, 2012.
- Akhlaghi, M., M. Ghasemi, and M. Riasatian, et al., Soy isoflavones prevent bone resorption and loss, a systematic review and meta-analysis of randomized controlled trials, Crit. Rev. Food Sci. Nutr. 1–15, 2019.
- Taku, K., M.K. Melby, and F. Kronenberg, et al., Extracted or synthesized soybean isoflavones reduce menopausal hot flash frequency and severity: systematic review and meta-analysis of randomized controlled trials, Menopause 19:776–90, 2012.
- Kumar, V., A.K. Sinha, and H.P.S. Makkar, et al., Dietary roles of phytate and phytase in human nutrition: a review, Food Chem.1209: 45–59, 2010.
- Kunyanga, C.N., J.K. Imungi, and M.W. Okoth, et al., Antioxidant and type-2-diabetes- related functional properties of phytic acid extract from Kenyan local food ingredients: effects of traditional processing methods, Ecol. Food Nutr. 50 :452–71, 2011.
- Clemente, A. and M.D. Arques, Bowman-Birk inhibitors from legumes as colorectal chemopreventive agents, World journal of gastroenterology 20: 10305–15, 2014.
- Yamamoto, S., M. Tomiyama, and R. Nemoto, et al., Effects of food lectins on the transport system of human intestinal Caco-2 cell monolayers, Biosci. Biotechnol. Biochem. 77: 1917–24, 2013.
- Muramoto, K., Lectins as bioactive proteins in foods and feeds, Food Sci. Technol. Res. 23: 487–94, 2017.
- Ryan, C.A., Proteinase inhibitors in plants: genes for improving defenses against insects and pathogens, Annu. Rev. Phytopathol. 28: 425-49, 1990.
- Aviles-Gaxiola, S., C. Chuck-Hernandez, and S.O. Serna Saldivar, Inactivation methods of trypsin inhibitor in legumes: a review, J. Food Sci. 83: 17–29, 2018.
- Habib, H. and K.M. Fazili, Plant protease inhibitors: a defense strategy in plants, Biotechnol. Mol. Biol. Rev. 2: 268–85, 2007.
- Srikanth, S. and Z. Chen, Plant protease inhibitors in therapeutics-focus on cancer therapy, Front. Pharmacol. 7: 470, 2016.
- Shi, L., K. Mu, and S.D. Arntfield, et al., Changes in levels of enzyme inhibitors during soaking and cooking for pulses available in Canada, J. Food Sci. Technol. 54: 1014–22, 2017.
- Khattab, R.Y. and S.D. Arntfield, Nutritional quality of legume seeds as affected by some physical treatments 2. Antinutritional factors, Food Sci. Technol. 42: 1113–8, 2009.
- Vats, C. and U.S. Banerjee, Production studies and catalytic properties of phytases (myo-inositol-hexakis-phosphate phosphohydrolases): an overview, Enzyme Microbial Technol. 35: 3–14, 2004.
- Reddy, N.R. and S.K. Sathe, Phytases in legumes and cereals, Adv. Food Res. 28:1–92, 1982.
- Prynne, C.J., A. McCarron, and M.E. Wadsworth, et al. Dietary fiber and phytate—a balancing act: results from three time points in a British birth cohort, Br. J. Nutr. 103: 274–80, 2010.
- Schlemmer, U., W. Frolich, and R.M. Prieto, et al., Phytate in foods and significance for humans: food sources, intake, processing, bioavailability, protective role, and analysis, Mol. Nutr. Food Res. 53 Suppl 2S330–75, 2009.
- Gibson, R.S., V. Raboy, and J.C. King, Implications of phytate in plant-based foods for iron and zinc bioavailability, setting dietary requirements, and formulating programs and policies, Nutr. Rev. 76: 793–804, 2018.
- Campos-Vega, R., G. Loarca-Piña, and B.D. Oomah, Minor components of pulses and their potential impact on human health, Food Res. Int. 43: 461–482, 2010.
- Shi, L., S.D. Arntfield, and M. Nickerson, Changes in levels of phytic acid, lectins, and oxalates during soaking and cooking of Canadian pulses, Food Res. Int. 107: 660–8, 2018.
- Institute of Medicine. Food and Nutrition Board. Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc : a Report of the Panel on Micronutrientsexternal link disclaimer. Washington, DC: National Academy Press; 2001.
- EFSA, Scientific opinion on Dietary Reference Values for zinc, EFSA J. 12: 38–44, 2014.
- Lynch, S.R., J.L. Beard, and S.A. Dassenko, et al., Iron absorption from legumes in humans, Am. J. Clin. Nutr. 40: 42–7, 1984.
- Petry, N., I. Egli, and B. Campion, et al., Genetic reduction of phytate in common bean (Phaseolus vulgaris L.) seeds increases iron absorption in young women, J. Nutr. 143: 1219–24, 2013.
- Heaney, R.P., C.M. Weaver, and M.L. Fitzsimmons, Soybean phytate content: effect on calcium absorption, Am. J. Clin. Nutr. 53(3):745-7, 1991.
- Weaver C.M., R.P. Heaney, and W.R. Proulx, et al., Absorbability of calcium from common beans, J. Food Sci. 58: 1401–3, 1993.
- Sandstrom, B., A. Almgren, and B. Kivisto, et al., Effect of protein level and protein source on zinc absorption in humans, J. Nutr. 119: 48–53, 1989.
- Cook, J.D., S.A. Dassenko, and S.R. Lynch, Assessment of the role of nonheme-iron availability in iron balance, Am. J. Clin. Nutr. 54: 717–22, 1991.
- Hambidge, K.M., J.W. Huffer, and V. Raboy, et al., Zinc absorption from low-phytate hybrids of maize and their wild-type isohybrids, Am. J. Clin. Nutr. 79: 1053–9, 2004.
- Brown, K.H., J.A. Rivera, and Z. Bhutta, et al., International Zinc Nutrition Consultative Group (IZiNCG) technical document #1. Assessment of the risk of zinc deficiency in populations and options for its control, Food and Nutrition Bulletin 25(1 Suppl 2): S99–203,2004.
- Brune, M., L. Rossander, and L. Hallberg, Iron absorption: no intestinal adaptation to a high-phytate diet, Am. J. Clin. Nutr. 49: 542–5, 1989.
- Armah, S.M., E. Boy, and D. Chen, et al., Regular consumption of a high-phytate diet reduces the inhibitory effect of phytate on nonheme-iron absorption in women with suboptimal iron stores, J. Nutr. 145: 1735–9, 2015.
- Murray-Kolb, L.E., R. Welch, and E.C. Theil, et al., Women with low iron stores absorb iron from soybeans, Am. J. Clin. Nutr. 77: 180–4, 2003.
- Lonnerdal, B., A. Bryant, and X. Liu, et al., Iron absorption from soybean ferritin in nonanemic women, Am. J. Clin. Nutr. 83: 103–7, 2006.
- Lonnerdal, B., Soybean ferritin: implications for iron status of vegetarians, Am. J. Clin. Nutr. 89: 1680S–5S, 2009.
- Hoppler, M., C. Zeder, and T. Walczyk, Quantification of ferritin-bound iron in plant samples by isotope tagging and species-specific isotope dilution mass spectrometry, Anal. Chem. 81: 7368–72, 2009.
- Liebman, M. and L. Doane, Calcium and zinc balances during consumption of high and low oxalate-containing vegetables, Nutr. Res. 9: 947–55, 1989.
- He,W., X. Li, and K. Ding, et al., Ascorbic acid can reverse the inhibition of phytic acid, sodium oxalate and sodium silicate on iron absorption in Caco-2 cells, Int. J. Vitam. Nutr. Res. 88: 65–72, 2018.
- genannt Bonsmann, S.S., T. Walczyk, and S. Renggli, et al., Oxalic acid does not influence nonhaem iron absorption in humans: a comparison of kale and spinach meals, Eur. J. Clin. Nutr. 62: 336–41, 2008.
- Kelsay, J.L. and E.S. Prather, Mineral balances of human subjects consuming spinach in a low-fiber diet and in a diet containing fruits and vegetables, Am. J. Clin. Nutr. 38: 12–9, 1983.
- Weaver, C.M., R.P. Heaney, and K.P. Nickel, et al., Calcium bioavailability from high oxalate vegetables: Chinese vegetables, sweet potatoes, and rhubarb, J. Food Sci. 63: 524–5, 1997.
- Zhao, Y., B.R. Martin, and C.M. Weaver, Calcium bioavailability of calcium carbonate fortified soymilk is equivalent to cow's milk in young women, J. Nutr. 135: 2379–82, 2005.
- Weaver, C.M., R.P. Heaney, and L. Connor, et al., Bioavailability of calcium from tofu vs. milk in premenopausal women, J. Food Sci. 683: 144–7, 2002.
- Romero, V., H. Akpinar, and D.G. Assimos, Kidney stones: a global picture of prevalence, incidence, and associated risk factors, Rev. Urol. 12: e86–96, 2010.
- Stamatelou, K.K., M.E. Francis, and C.A. Jones, et al., Time trends in reported prevalence of kidney stones in the United States: 1976–1994, Kidney Int. 63: 1817–23, 2003.
- Scales, C.D., A.C. Smith, and J.M. Hanley, et al., Prevalence of kidney stones in the United States, Eur. Urol. 62: 160–5, 2012.
- Daudon, M., R. Donsimoni, and C. Hennequin, et al., Sex- and age-related composition of 10 617 calculi analyzed by infrared spectroscopy, Urol. Res. 23: 319–26, 1995.
- Robertson, W.G. and M. Peacock, The cause of idiopathic calcium stone disease: hypercalciuria or hyperoxaluria? Nephron. 26: 105–10, 1980.
- Han H., A.M. Segal, and J.L. Seifter, et al., Nutritional management of kidney stones (nephrolithiasis), Clin. Nutr. Res. 4: 137–52, 2015.
- Chai, W. and M. Liebman, Oxalate content of legumes, nuts, and grain-based flours, J. Food Comp. Anal. 18: 723–29, 2005.
- Chai W. and M. Liebman, Effect of different cooking methods on vegetable oxalate content, J. Agric. Food Chem. 53: 30: 27–30, 2005.
- Jaworska, G., Content of nitrates, nitrites, and oxalates in New Zealand spinach, Food Chem. 89: 235–42, 2005.
- Savage, G.P., L. Vanhanen, and S.M. Mason, et al., Effect of cooking on the soluble and insoluble oxalate content of some New Zealand foods, J. Food Comp. Anal. 13: 201–6, 2000.
- Judprasong, K., S. Charoenkaitkul, and P. Sungpang, et al., Total and soluble oxalate contents in Thai vegetables, cereal grains, and legume seeds and their changes after cooking, J. Food Comp. Anal. 19: 340-7, 2006.
- Toma, R.B., M.M. Tabekhia, and J.D. Williams, Phytate and oxalate contents in sesame seed (Sesamum indicum l.), Nutr. Reports Int. 20: 25–31, 1979.
- Sorensen, M.D., Calcium intake and urinary stone disease, Transl. Androl. Urol. 3: 235–40, 2014.
- Tang, M., D.E. Larson-Meyer, and M. Liebman, Effect of cinnamon and turmeric on urinary oxalate excretion, plasma lipids, and plasma glucose in healthy subjects, Am. J. Clin. Nutr. 87(5):1262–7, 2008.
- Chai, W. and M. Liebman, Assessment of oxalate absorption from almonds and black beans with and without the use of an extrinsic label, J. Urol. 172: 953–7, 2004.
- Meschi, T., A. Nouvenne, and L. Borghi, Lifestyle recommendations to reduce the risk of kidney stones, The Urologic clinics of North America 38: 313–20, 2011.
- Robertson, W.G., M. Peacock, and D.H. Marshall, Prevalence of urinary stone disease in vegetarians, Eur. Urol. 8: 334–9, 1982.
- Taylor, E.N., T.T. Fung, and G.C. Curhan, DASH-style diet associates with reduced risk for kidney stones, J. Am. Soc. Nephrol. 20: 2253–9, 2009.
- Grases, F., R. Garcia-Gonzalez, and J.J. Torres, et al., Effects of phytic acid on renal stone formation in rats, Scand. J. Urol. Nephrol. 32: 261–5, 1998.
- Curhan, G.C., W.C. Willett, and E.L. Knight, et al., Dietary factors and the risk of incident kidney stones in younger women: Nurses' Health Study II, Arch. Intern. Med. 164: 885–91, 2004.
- Champ, M.M., Non-nutrient bioactive substances of pulses, Br. J. Nutr. 88 Suppl. 3: S307–19, 2002.
- Gold, E.R. and P. Balding. Receptor-Specific Proteins: Plant and Animal Lectins. New York: American Elsevier Publishing Co.; 1975.
- Sharon, N. and H. Lis, History of lectins: from hemagglutinins to biological recognition molecules, Glycobiol. 14: 53R–62R, 2004.
- Rudiger, H., Plant lectins—more than just tools for glycoscientists: occurrence, structure, and possible functions of plant lectins, Acta Anat. (Basel)161: 130–52, 1998.
- Liener, I.E., Soyin, a toxic protein from the soybean: I. Inhibition of rat growth, J. Nutr. 49 529–39, 1953.
- Pusztai, A. and S. Bardocz, Biological effects of plant lectins on the gastrointestinal tract: metabolic consequences and applications, Trends Glycosci. Glyc. 8: 149–65, 1996.
- Peumans, W.J. and E.J. Van Damme, Lectins as plant defense proteins, Plant Physiol. 109: 347–52, 1995.
- Hirsch, A.M., Role of lectins (and rhizobial exopolysaccharides) in legume nodulation, Curr. Opin. Plant Biol. 2: 320–6, 1999.
- Banwell, J.G., R. Howard, and D. Cooper, et al., Intestinal microbial flora after feeding phytohemagglutinin lectins (Phaseolus vulgaris) to rats, Appl. Environ. Microbiol. 50: 68–80, 1985.
- Baintner, K., G. Jakab, and Z. Gyori, et al., Binding of FITC-labelled lectins to the gastrointestinal epithelium of the rat, Pathol. Oncol. Res. 6: 179–83, 2000.
- Vasconcelos, I.M. and J.T. Oliveira, Antinutritional properties of plant lectins, Toxicon. 44: 385–403, 2004.
- Wang, Q., L.G. Yu, and B.J. Campbell, et al., Identification of intact peanut lectin in peripheral venous blood, Lancet 352: 1831–2, 1998.
- Noah, N.D., A.E. Bender, and G.B. Reaidi, et al., Food poisoning from raw red kidney beans, Br. Med. J. 281: 236–7, 1980.
- Bender, A.E. and G.B. Reaidi, Toxicity of kidney beans (Phaseolus vulgaris) with particular reference to lectins, J. Plant Foods 4: 15–22, 1982.
- Gilbert, R.J., Healthy eating day, Commun. Dis. Rep. 3: 33–4, 1988.
- Rodhouse, J.C., C.A. Haugh, and D. Roberts, et al., Red kidney bean poisoning in the UK: an analysis of 50 suspected incidents between 1976 and 1989, Epidemiol. Infect. 105: 485–91, 1990.
- Ogawa, H. and K. Date, The "white kidney bean incident" in Japan, Methods Mol. Biol. 1200: 39–45, 2014.
- Audi, J., M. Belson, and M. Patel, et al., Ricin poisoning: a comprehensive review, JAMA 294: 2342–51, 2005.
- Freed, D.L., Do dietary lectins cause disease? BMJ 318: 1023–4, 1999.
- Pusztai, A., S.W. Ewen, and G. Grant, et al., Relationship between survival and binding of plant lectins during small intestinal passage and their effectiveness as growth factors, Digestion. 46 Suppl. 2: 308–16, 1990.
- Machado, F.P.P., J.H. Queiroz, and M.G.A.Oliveira, et al., Effects of heating on protein quality of soybean flour devoid of Kunitz inhibitor and lectin, Food Chem. 107: 649–55, 2008.
- Pusztai, A., S.W. Ewen, and G. Grant, et al., Lectins and also bacteria modify the glycosylation of gut surface receptors in the rat, Glycoconj. J. 12: 22–35, 1995.
- Banwell, J.G., D.H. Boldt, and J. Meyers, et al., Phytohemagglutinin derived from red kidney bean (Phaseolus vulgaris): a cause for intestinal malabsorption associated with bacterial overgrowth in the rat, Gastroenterology 84: 506–15, 1983.
- Boyd, W.C. and R.M. Reguera, Hemagglutinating substances for human cells in various plants, J Immunol. 62: 333-9, 1949
- Brücher, O., M. Wecksler, and A. Levy, et al., Comparison of phytohaemagglutinins in wild beans (Phaseolus aborigineus) and in common beans (Phaseolus vulgaris) and their inheritance, Phytochemistry 8: 1739–43, 1969.
- Pull, S.P., S.G. Pueppke, and T. Hymowitz, et al., Soybean lines lacking the 120,000-dalton seed lectin, Science 200: 1277–9, 1978.
- Armour, J.C., R.I.C. Chanaka, and W.C. Buchan, et al., Protease inhibitors and lectins in soybeans and effects of aqueous heat-treatment, J. Sci. Food Agric. 78: 225–31, 1998.
- Grant, G., L.J. More, and N.H. McKenzie, et al., The effect of heating on the haemagglutinating activity and nutritional properties of bean (Phaseolus vulgaris) seeds, J. Sci. Food Agric. 33: 1324–6, 1982.
- Grant, G., van Drtessche E. Legume lectins. physicochemlcal and nutritional properties. In: Recent Advances of Research in Ant-nutritional Factors in Legume Seeds (1993) eds. van der Poel AFB, Huisman J, Sam HS, Wagenmgen (pp. 219–34) The Netherlands: Wagenmgen Press.
- Nachbar, M.S. and J.D. Oppenheim, Lectins in the United States diet: a survey of lectins in commonly consumed foods and a review of the literature, Am. J. Clin. Nutr. 33: 2338–45, 1980.
- Uchigata, Y., S.L. Spitalnik, and O. Tachiwaki, et al., Pancreatic islet cell surface glycoproteins containing Gal beta 1-4GlcNAc-R identified by a cytotoxic monoclonal autoantibody, J. Exp. Med. 165: 124–39, 1987.