Two advances in biodiesel technology
By P.M. Nielsen, William Summers
July/August 2014
This issue of Inform features two advances in biodiesel technology—one based on a liquid lipase and the other on solid catalysts.
Enzyme-catalyzed biodiesel made from low-quality oils
- In the first quarter of 2014, both Blue Sun Biodiesel in St. Joseph, Missouri, USA, and Vieselfuel LLC in Stuart, Florida, USA, announced the full-scale production of biodiesel based on lipase as catalyst.
- Production at both sites has been in operation for over a year now. Novozymes has been the enzyme supplier and partner, and the accomplishment of full-scale production is the result of lengthy, dedicated research and development work.
- The new lipase technology enables the processing of oil feedstocks with any concentration of free fatty acids and with lower energy costs than with a standard chemical catalyst. In this article, a senior scientist manager at Novozymes describes the process and how it was developed.
Utilizing lipases in the production of biodiesel dates back more than 10 years, and a considerable number of articles suggest the use of immobilized enzymes (Fjerbaek, L., et al., 2009). The first trials using liquid formulated lipases instead of immobilized took place at Novozymes’ laboratories in 2006 and resulted in the first patent filings.
In 2008, the Danish National Advanced Technology Foundation supported a large research effort involving universities and a biodiesel producer. At the same time, Novozymes began a collaboration with Piedmont Biofuels in Pittsboro, North Carolina, USA. The objectives of both projects were to find a lipase with a selling price low enough to compete in the chemical biodiesel market and to demonstrate the enzymatic biodiesel process in pilot or production scale. Originally, the collaborators believed that the result would be a low-cost immobilized lipase, but with time the most efficient process proved to be one with a new liquid formulated lipase (Cesarini, S., et al., 2013). The results led to the latest patent filing in 2012, which describes the basis for the BioFAME® process utilizing liquid-formulated lipases as a catalyst and includes the reuse of the enzyme (Patent WO2012/098114, 2012).
The final enzymatic biodiesel process consists of an enzyme reaction step followed by polishing as shown in Figure 1.
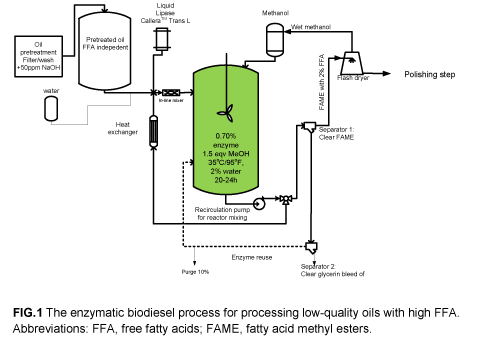
The operating principle of the enzyme reactor is the creation of an emulsion with a small amount of water (1–2%), as the enzyme works specifically at the interface between oil and water. Constant and efficient mixing during the reaction is required. One crucial specification for the oil feedstock was discovered; it must not contain acidity from mineral acids added upstream. Neutralization of such acids can be ensured by, for instance, 50 ppm NaOH added as a 10% solution. The reaction temperature must be controlled to 35°C/95°F, and the methanol added gradually to prevent enzyme inactivation. Typically, the required methanol is added during the first 6–10 hours of reaction. An efficient enzyme dosage of 0.7% is suggested, and with the reuse option the enzyme consumption will be close to 0.2% w/w on oil. It is only in the first batch that the addition of water is required. During additional batches the water from the reused heavy phase and the wet methanol is normally sufficient.
Figure 1 shows the reactor in connection with centrifuges to separate the fatty acid methyl esters (FAME) and glycerin after the reaction. Alternatively, gravity settling in the reactor can be used, but it requires a relatively long time to produce clear glycerin. In either case, a small loss of enzyme activity occurs in every batch. The methanol/temperature conditions cause a slight inactivation of the enzyme, and there is a physical loss of enzyme in the separation step. Experience can ensure that the overall enzyme activity loss is limited to <15% per batch.
Use of the liquid lipases was a breakthrough, as they are much cheaper to produce and provide technological as well as cost benefits. By using the lipase Novozymes Callera Trans®, it is possible to produce biodiesel from a large variety of oil qualities. The ability to produce biodiesel from feedstock regardless of its FFA content ultimately makes the process a more cost-efficient way to produce biodiesel.
One of the key technologies involved is the recovery of the enzyme. The reaction time of 20–24 hours is dependent on a certain concentration of enzyme, for example, 0.7% of the oil. To lower associated costs, the enzyme is collected and reused. After the reaction, the reaction mixture is separated by gravity/centrifuge into three layers as illustrated in Figure 2. The glycerin phase after separation is very different from the glycerin obtained from an alkaline-catalyzed process, as it is almost free from salt.

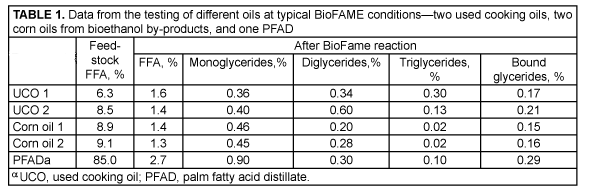
The FAME phase from the enzyme reaction typically consists of a composition with bound glycerin <0.22% and FFA 2%. The FFA content varies, as it is dependent on the FFA content in the feed. At very high FFA content, such as that found in palm fatty acid distillate, it can typically reach 2.5–3.0% FFA. A low FFA content after the reaction can be achieved by controlling the water and methanol contents, taking the water formed by the FFA esterification also into consideration. Data from different oil reactions are included in Table 1.
The polishing step is required mainly owing to the FFA content which has to be reduced to <0.25% according to ASTM specification. This can take place as one of several alternative process steps:
1. Caustic wash. The caustic wash is based on the refining concept that eliminates FFA by a NaOH wash of virgin oil. The residual FFA content in the FAME phase is relatively low and the formation of soap is limited. However, the solubility of soap in the FAME is different from its solubility in oil, and a higher recirculation volume of soap/FAME than the normal 2.5 times soap volume is required. One benefit of the caustic wash is the significant reduction in monoglycerides.
2. Resin esterification. Resin technology is used today to eliminate FFA from oil as a pretreatment to biodiesel production with Na-methoxide catalyst. The concept is also applicable as a polishing step and uses a resin catalyzing the esterification at high temperatures (90°C/195°F) and methanol concentration (15–20%).
3. Sulfuric acid esterification. The sulfuric acid esterification is well established as a pretreatment for high-FFA feedstocks, for example, animal fat. There are limitations to the level of FFA that can be esterified, and the equipment has to be glass lined to prevent excessive corrosion. As the BioFAME reaction delivers FFA at a typical 2%, the sulfuric acid process might be able to reach in-specification FFA levels in one step.
4. Enzymatic esterification. Technically, this is probably the most advantageous of the processes mentioned. Aside from the FFA esterification, it also ensures the transesterification of the remaining glycerides. The cost of the enzyme needs to be considered in this case.
Distillation of the final product is an option to secure against any carryover from low -quality oils, for example, to ensure that waxes or metal ions are not found in the final biodiesel. An improved color and cold soak quality can also be secured by distillation.
Novozymes is currently finalizing the development work of the enzymatic biodiesel application and is ready to officially launch the concept later this year. Together with our partners who are using the lipase Callera Trans in full-scale production, we have shown that biodiesel can be produced from oils having different low qualities independent of FFA content and having a low cost for methanol recovery. The process has been installed at two full-scale plants, one as a retrofitted process to a traditional plant and the other as a greenfield plant. This is the first step into the biodiesel industry, but future perspectives for enzymatic processes are already foreseen, such as combined degumming and transesterification and sterylglycoside acylation.
P.M. Nielsen is senior science manager with Novozymes R&D group Bioenergy Opportunities.Contact: pmn@novozymes.com.
Information
Cesarini, S., P. Diaz, and P.M. Nielsen, Exploring a new, soluble lipase for FAMEs production in water-containing systems using crude soybean oil as a feedstock, Process Biochem. 48:484–487, 2013.
Fjerbaek, L., K.V. Christensen, and B. Norddahl, A review of the current state of biodiesel production using enzymatic transesterification, Biotechnol. Bioeng. 102:1298–1315, 2009.
Nielsen, P.M., Production of fatty acid alkyl esters, World Intellectual Property Organization Patent WO2012/098114, 2012.
Innovative catalysts open new opportunities in biodiesel market
A new proprietary solid catalyst process developed by Benefuel:
- converts inedible fats and oils, which are renewable feedstocks, into specification biodiesel;
- converts both glycerides and free fatty acids into alkyl esters for industrial use; and
- operates at lower cost than any other current process for esterification or transesterification.
The current, widely used process for producing biodiesel dates to chemistries extant since the late 19th and early 20th centuries: batch operations, using homogeneous catalysts, batch separations, and the like. As the US biodiesel industry began to expand rapidly in 2005, US producers faced the persistent problems of competing with the food industry for the same feedstock—refined, bleached, and deodorized (RBD) soybean oil—and ever-increasing prices and thinner margins. Along with this came the still-simmering “food vs. fuel” debate over how US agricultural potential should be allocated.
While the opportunity for a new biodiesel technology capable of processing non-food feedstock was vast, to be commercially viable such a technology had to be better suited to large-volume commodity production, to be largely insensitive to feedstock variability, and to provide a better return on invested capital than existing technologies. For Benefuel (Irving, Texas, USA), the challenge was to surpass the initial entry of Axens (Salindres, France) Esterfip-H® catalyst, which entered the US market in 2007.
In working with our partners at the National Chemical Laboratory (NCL; Pune, India), a new solid powder catalyst capable of both esterification and transesterification emerged. This new catalyst afforded high yields of fatty acid methyl esters (FAME) and glycerin under mild conditions (Sreeprasanth et al., 2006) using a wide variety of available fats and oils. Free fatty acids (FFA), which are common in less refined and less expensive feedstocks such as poultry fat, yellow grease, and palm oil derivatives, have long posed serious problems in conventional biodiesel processing. Benefuel licensed the exclusive worldwide rights to this NCL technology in 2006 and continued discovery and development work with NCL.
Within the first year after signing the license agreement, Benefuel had developed with NCL a second solid catalyst—more suited to fixed-bed applications and thus large-scale commercial fuel production—to accompany the first powder catalyst, which is highly effective in batch operation. Both catalysts are effective in converting fatty acids (FA), fats or oils, and mixtures of these into methyl esters. Benefuel began work on process scale-up in fixed-bed reactors in 2008.
To us, the path ahead was clear: The biodiesel industry needed a fully continuous, fully integrated production refinery for biodiesel—one that could receive a variety of feedstocks and process them continuously to biodiesel and glycerin. The fixed-bed reactor design and our new catalyst were at the heart of this approach. Although the wide versatility of our catalysts for esterification and transesterification were well recognized, development of other applications had to wait for process validation in biodiesel.
Benefuel’s Ensel® fixed-bed process is quite simple (Fig.1). It employs our second solid catalyst, which was developed in conjunction with Süd-Chemie India Pvt. Ltd. (Kerala)and patented in the United States and Japan (US 8,124,801 and JP 5,470,382) with applications in other countries.
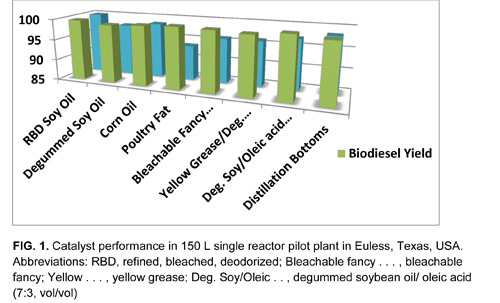
This durable, promoted, metal oxide catalyst is largely insensitive to water and effectively converts every feedstock that has been tested in numerous pilot plant-scale operations. Examples include degummed soybean oil, cottonseed oil, corn oil from dried distillers’ grains with solubles, yellow grease, beef tallow, crude palm oil, palm fatty acid distillate, and even a mixture of degummed soybean oil and oleic acid (7:3, vol/vol).
The basic design of Benefuel’s ENSEL process for transesterification involves three major components: fixed-bed reactors, an oil–glycerin separation stage, and a pair of distillation columns. Each element of the process operates continuously and can be monitored at critical points with inline sensors for tight control.
The process starts with renewable, inedible feedstock, requiring some minimal pretreatment to remove insolubles and water, which would otherwise displace feedstock. The catalyst is contacted with methanol and feedstock under specific conditions of temperature (190–210°C), pressure (40–50 bar) , and flow rate (weight hourly space velocity = 0.4–0.6/hr), followed by recovery and refinement of the excess methanol, product separation, and FAME distillation. Still bottoms can be recycled to increase carbon efficiency, and the glycerin co-product is low in both ash and non-glycerol organic matter.
The reactor is generally columnar in shape, suited for medium-pressure service at moderate temperatures in flooded mode. In this vessel, the reagents—triglycerides (TG) and an excess of methanol in the liquid phase—come in contact with the active catalyst’s surface, which accelerates the transformation of the glycerides to methyl esters.
As the liquid stream exits the reactor, the pressure is reduced on the stream of crude biodiesel, and methanol and the volatile methanol (and water if the feedstock contains FFA) quickly flash to vapor and are carried directly to the methanol refining distillation unit.
The separation can be a simple decanter, in which the product crude biodiesel (the oil layer) and glycerin mixture separates because of the difference in density. Benefuel also holds exclusive rights in a novel electrostatically enhanced separation system, which can dramatically shorten the residence time compared to conventional decanters.
The recovered glycerin can be pumped to a small vacuum distillation column to remove any volatiles (3–4% of total glycerin volume) and then to co-product storage. The volatiles, consisting mostly of water and methanol, are pumped to the methanol recovery system.
Distillation of the recovered oil phase is the last stage of the process. This two-step distillation removes any residual volatiles (first stage: residual methanol, water, and volatile unsaponifiables) and refines the methyl esters from any higher-boiling impurities (second stage: unconverted glycerides and high-boiling unsaponifiables). The recovered methyl esters are continuously analyzed against ASTM specifications for B100 validation.
Distillation of the crude biodiesel ensures continuous, high-quality output and minimal risk of cold flow issues caused by residual glycerides. As specifications continue to tighten, even conventionally designed biodiesel plants are adding a final stage distillation.
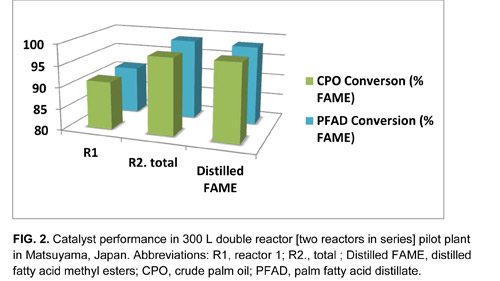
In 2009, as work on the biodiesel process scale-up shifted from NCL’s labs to Benefuel’s domestic and Japanese pilot plants (Fig. 2), work at NCL refocused to develop other reaction modes with these same catalysts. Among these are the conversion of TG or fatty acids into alkyl fatty acid esters with higher-boiling alcohols and of FAME and acyl glycerides into polyol esters, for which we now have three separate process modes: batch (stirred tank reactor), continuous (fixed bed reactor or catalytic reactive distillation). These processes leading to biodegradable lubricant base oil and other oleochemicals await pilot-scale testing for commercial applications.
The solid catalyst advantage
The advantages of solid catalytic processing are simply economic—continuous production at commercial scale without reference to periodic changes of feedstock or feedstock blends; a catalyst life of several years instead of “catalyst as reagent,” as in conventional biodiesel production; and an ability to blend feedstocks to achieve optimal cold-flow properties in the final product at a low raw material cost.

When compared on the basis of production, Benefuel estimates that the ENSEL process has the lowest cost producer advantage in the market (Fig. 3). This competitive advantage offers ENSEL producers a strong economic edge over conventional biodiesel production processes in their numerous variations. Except for “green” diesel, which requires a source of hydrogen and affords no glycerin co-product, ENSEL can be adapted to existing and greenfield operations.
Benefuel is currently partnered with Flint Hills Resources LLC (Wichita, Kansas, USA) in the retrofitting of the former Axens’ biodiesel plant in Beatrice, Nebraska, USA to operate with the ENSEL process and is engaged in one other US greenfield biodiesel project, while pursuing other opportunities in Southeast Asia and Canada.
William Summers is chief science officer of Benefuel Inc. Since 1974, Summers has managed multiple technology businesses with a focus on developing processes for new products and improving existing operational efficiencies. He can be contacted at wsummers@benefuel.net.
Sidebar
The advantages of using solid acid catalysts
Solid acid catalysts offer several advantages for continuous or batch operation production of biodiesel and synthetic esters. These include:
- no feedstock constraints; can use fats, oils, methyl esters, or fatty acids
- wide range of feedstock quality choices
- limited pretreatment needed, to remove insolubles
- lower capital expenditures (CAPEX) and operating expenditures (OPEX) than conventional processes
- adaptable to “greenfield,” “brownfield,” or “retrofit” facilities
- lowest cost of production of fatty acid methyl esters
- nonaqueous downstream processing, minimal waste streams
- better conversion and higher selectivity for esters <bl>
Information
Sreeprasanth, P.S., R. Srivastava, D. Srinivas, and P. Ratnasamy, Hydrophobic, solid acid catalysts for production of biofuels and lubricants, Appl. Catal. A: General 314:148–159, 2006.
Srinivas, D., P. Rattnassamy, S.A. Pardhy, T. Raja, S.S. Deshpande, et al., Process of manufacturing of fatty acid alkyl esters, US Patent 8124801 and Japanese Patent 5470382, Benefuel Inc., February 28, 2012.
